Animal NDC 13985-726-16 Vetraseb M
Chlorhexidine Gluconate, Miconazole Nitrate, Phytosphingosine Salicyloyl
Animal Product Information
| Field Name | Field Value |
|---|---|
| Animal NDC Code | 13985-726-16 |
| Proprietary Name | Vetraseb M What is the Proprietary Name? The proprietary name also known as the trade name is the name of the product chosen by the medication labeler for marketing purposes. |
| Non-Proprietary Name | Chlorhexidine Gluconate, Miconazole Nitrate, Phytosphingosine Salicyloyl What is the Non-Proprietary Name? The non-proprietary name is sometimes called the generic name. The generic name usually includes the active ingredient(s) of the product. |
| Labeler Name | Mwi/vetone |
| Product Type | Otc Animal Drug |
| Usage Information |
|
| Active Ingredient(s) |
|
| Inactive Ingredient(s) |
|
| Marketing Category | UNAPPROVED DRUG OTHER What is the Marketing Category? Product types are broken down into several potential Marketing Categories, such as NDA/ANDA/BLA, OTC Monograph, or Unapproved Drug. One and only one Marketing Category may be chosen for a product, not all marketing categories are available to all product types. Currently, only final marketed product categories are included. The complete list of codes and translations can be found at www.fda.gov/edrls under Structured Product Labeling Resources. |
Vetraseb M Images
Vetraseb M Animal Product Labeling Information
The product labeling information includes all published material associated to a drug. Product labeling documents include information like generic names, active ingredients, ingredient strength dosage, routes of administration, appearance, usage, warnings, inactive ingredients, etc.
Table of Contents
Description
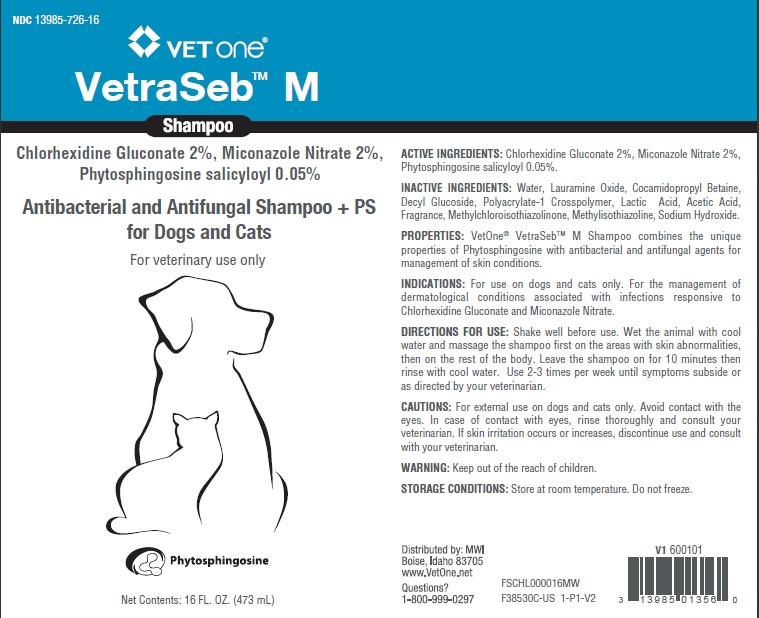
VetraSeb™ M SHAMPOO
ACTIVE INGREDIENTS: Chlorhexidine Gluconate 2%, Miconazole Nitrate 2%, Phytosphingosine salicyloyl 0.05%.
INACTIVE INGREDIENTS: Water, Lauramine Oxide, Cocamidopropyl Betaine, Decyl Glucoside, Polyacrylate-1 Crosspolymer, Lactic Acid, Acetic Acid, Fragrance, Methylchloroisothiazolinone, Methylisothiazoline, Sodium Hydroxide.
PROPERTIES: VetOne® VetraSebTM M Shampoo combines the unique properties of Phytosphingosine with antibacterial and antifungal agents for management of skin conditions.
Indications & Usage
INDICATIONS: For use on dogs and cats only. For the management of dermatological conditions associated with infections responsive to
Chlorhexidine Gluconate and Miconazole Nitrate.
DIRECTIONS FOR USE: Shake well before use. Wet the animal with cool water and massage the shampoo first on the areas with skin abnormalities, then on the rest of the body. Leave the shampoo on for 10 minutes then rinse with cool water. Use 2-3 times per week until symptoms subside or as directed by your veterinarian.
Warnings And Precautions
CAUTIONS: For external use on dogs and cats only. Avoid contact with the eyes. In case of contact with eyes, rinse thoroughly and consult your
veterinarian. If skin irritation occurs or increases, discontinue use and consult with your veterinarian.
WARNING: Keep out of the reach of children.
Storage And Handling
STORAGE CONDITIONS: Store at room temperature. Do not freeze.
Other
Distributed by: MWI
Boise, Idaho 83705
www.VetOne.net
Questions?
1-800-999-0297
Principal Display Panel - 8 Ounce (237 Ml) Bottle
NDC 3985-726-08
VETONE®
VetraSeb™ M
Shampoo
Chlorhexidine Gluconate 2%, Miconazole Nitrate 2%,
Phytosphingosine salicyloyl 0.05%
Antibacterial and Antifungal Shampoo +PS
for Dogs and Cats
For veterinary use only
Phytosphingosine
Net Contents: 8 FL. OZ. (237 mL)
Principal Display Panel - 16 Ounce (473 Ml) Bottle
NDC 3985-726-16
VETONE®
VetraSeb™ M
Shampoo
Chlorhexidine Gluconate 2%, Miconazole Nitrate 2%,
Phytosphingosine salicyloyl 0.05%
Antibacterial and Antifungal Shampoo +PS
for Dogs and Cats
For veterinary use only
Phytosphingosine
Net Contents: 16 FL. OZ. (473 mL)
* The information on this page is for an ANIMAL PRODUCT, please review the complete disclaimer below.