Animal NDC 50989-888-15 Hydravol Iv
Hydroxyethyl Starch, Sodium Chloride
Animal Product Information
Hydravol Iv Images
Hydravol Iv Animal Product Labeling Information
The product labeling information includes all published material associated to a drug. Product labeling documents include information like generic names, active ingredients, ingredient strength dosage, routes of administration, appearance, usage, warnings, inactive ingredients, etc.
Table of Contents
Description
HYDRAVOL IV™ (6% hydroxyethyl starch 130/0.4 in 0.9% sodium chloride injection) is a sterile, non-pyrogenic solution indicated for the treatment and prophylaxis of hypovolemia. It is not a substitute for red blood cells or coagulation factors in plasma. May be administered via intravenous infusion using aseptic technique. It contains no antimicrobial agents.
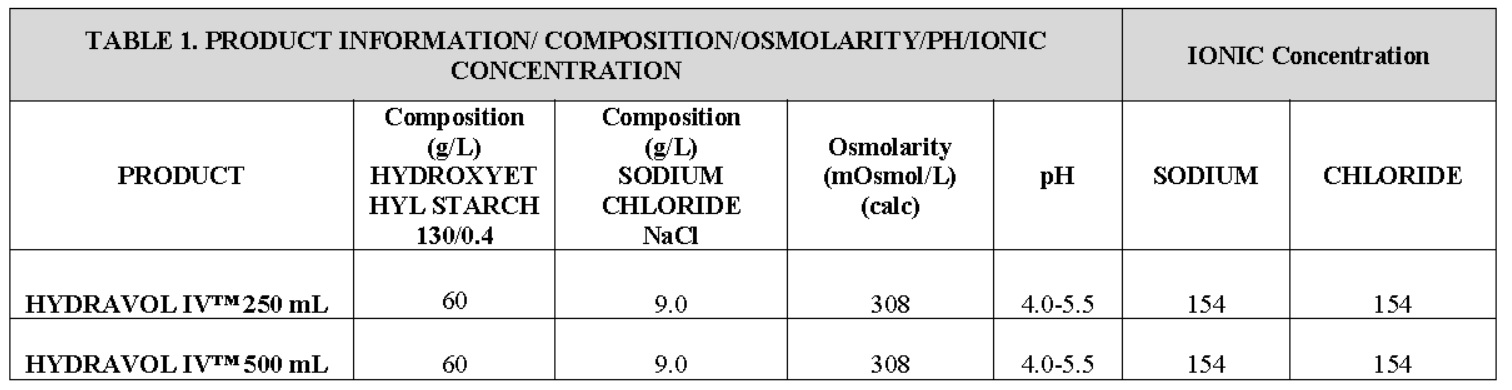
Composition, Osmolarity, pH, Ionic Concentration HYDRAVOL IV™ 250 mL and 500 mL:
The container is free of PVC and phthalates
Clinical Pharmacology
HYDRAVOL IV™ contains hydroxyethyl starch in a colloidal solution which expands plasma volume when administered intravenously. Hydroxyethyl starch is a derivative of thin boiling waxy corn starch, which mainly consists of a glucose polymer (amylopectin). Substitution of hydroxyethyl groups on the glucose units of the polymer reduces the normal degradation of amylopectin by α-amylase in the body.
Indications
HYDRAVOL IV™ acts as plasma volume substitute for the treatment and prophylaxis of hypovolemia. It is not a substitute for red blood cells or coagulation factors in plasma.
Contraindications
The use of HYDRAVOL IV™ is contraindicated in the following conditions:
- Known hypersensitivity to hydroxyethyl starch.
- Fluid overload (hyperhydration) and especially in cases of pulmonary edema and congestive heart failure.
- Renal failure with oliguria or anuria not related to hypovolemia.
- Patients receiving dialysis treatment.
- Severe hypernatremia or severe hyperchloremia.
- Intracranial bleeding.
Warnings
Anaphylactoid reactions (bradycardia, tachycardia, bronchospasm, non-cardiac pulmonary edema) have been reported with solutions containing hydroxyethyl starch. If a hypersensitivity reaction occurs, administration of the drug should be discontinued immediately, and the appropriate treatment and supportive measures should be undertaken until symptoms have resolved.
Fluid status and rate of infusion should be assessed regularly during treatment, especially in patients with cardiac insufficiency or severe kidney dysfunction.
In cases of severe dehydration, a crystalloid solution should be given first. Generally, sufficient fluid should be administered in order to avoid dehydration.
Caution should be observed before administering HYDRAVOL IV™ to patients with severe liver disease or severe bleeding disorders. With the administration of certain hydroxyethyl starch solutions, disturbances of blood coagulation can occur depending on the dosage.
If administered by pressure infusion, air should be withdrawn or expelled from the bag through the administration port prior to infusion.
Do not introduce additives into this container.
Adverse Reactions
- Products containing hydroxyethyl starch may lead to Anaphylactoid/hypersensitivity reactions.
- Prolonged administrations of high dosages of Hydroxyethyl starch may cause prutirus (itching), hemodilution (resulting in dilution of blood components, e.g., coagulation factors and other plasma proteins, and in a decrease in hematocrit).
- If an adverse reaction does occur, discontinue the infusion and evaluate the patient, institute appropriate therapeutic countermeasures and save the remainder of the fluid for examination, if deemed necessary.
Precautions
- Do not administer unless solution is clear and seal is intact.
- This is a single dose unit. It contains no preservatives.
- Use entire contents when first opened.
- Clinical evaluation and periodic laboratory determinations are necessary to monitor changes in fluid balance, electrolyte concentrations, and acid base balance during prolonged parenteral therapy, or whenever the patient's condition warrants such evaluation.
Drug Interactions
No interactions with other drugs or nutritional products are known. The safety and compatibility of additives have not been established.
Dosage And Administrations
- To be used as directed by a licensed veterinarian. HYDRAVOL IV™ is administered by intravenous infusion only. The daily dose and rate of infusion depend on the patient’s blood loss, on the maintenance or restoration of hemodynamics and on the hemodilution (dilution effect).
- For use in one patient on one occasion only. Discard any unused portion. Care should be taken with administration technique to avoid administration site reactions and infection.
- HYDRAVOL IV™ can be administered repetitively over several days. The initial 10 to 20 mL should be infused slowly, keeping the patient under close observation due to possible anaphylactoid reactions. See Warnings and Precautions.
- As a general recommendation, the class of synthetic colloids are prescribed at doses up to 20 mL per kg of body weight per day in small animal patient1. In a 30 kg patient, this is a dose of 600 mL of HYDRAVOL IV™ (equivalent to 1.2 g hydroxyethyl starch and 3.1 mEq sodium per kg of body weight).
ADULT DOSE
Overdosage
As with all plasma volume substitutes, overdosage can lead to overloading of the circulatory system (e.g., pulmonary edema). In this case, the infusion should be stopped immediately and, if necessary, a diuretic should be administered. See Warnings and Precautions.
Directions For Use Of Plastic Container
To open
Tear overwrap at slit and remove solution container. Some opacity of the plastic due to moisture absorption during the sterilization process may be observed. This is normal and does not affect the solution quality or safety. The opacity will diminish gradually. Check for minute leaks by squeezing inner bag firmly. If leaks are found, discard solution as sterility may be impaired.
Preparation for administration
- Suspend container from eyelet support.
- Remove plastic protector from inlet/outlet port at bottom of container.
- Attach administration set.
Warning: Do not introduce additives into this container.
Storage
Store at 15°C to 25°C (59°F to 77°F). Do not freeze.
How Supplied
HYDRAVOL IV™ (6% hydroxyethyl starch 130/0.4 in 0.9% sodium chloride injection) for intravenous infusion is supplied in the following primary container:
Polyolefin bag with overwrap: 250 mL and 500 mL
Manufactured For:
Vedco, Inc.
5503 Corporate Dr.
St Joseph, MO 64507 USA
For a Copy of the Safety Data Sheet (SDS) or to report adverse reactions call Vedco, Inc. customer service 1 (888) 708-3326
References
- Silverstein D, Hopper K. Small Animal Critical Care Medicine. (2009)
Principal Display Panel
NDC 50989-888-15
HydraVol IV™
6% Hydroxyethyl Starch 130/0.4 in 0.9% Sodium Chloride Injection
FOR INTRAVENOUS INFUSION ONLY
250mL
NDC 50989-888-16
HydraVol IV™
6% Hydroxyethyl Starch 130/0.4 in 0.9% Sodium Chloride Injection
FOR INTRAVENOUS INFUSION ONLY
500mL
* The information on this page is for an ANIMAL PRODUCT, please review the complete disclaimer below.