Product Images Alinia
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 10 images provide visual information about the product associated with Alinia NDC 67546-212 by Romark Laboratories, L.c., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
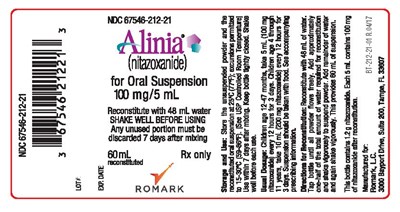
Alinia for Oral Suspension- Container Label - CROPPED BT 212 21 01 R.04 17 FINAL PROOF Alina 100mg 5mL OS Label
Alinia Tablet - 30 ct carton - CT 111 12 01 FINAL PROOF Alinia Tablets 30 ct Carton
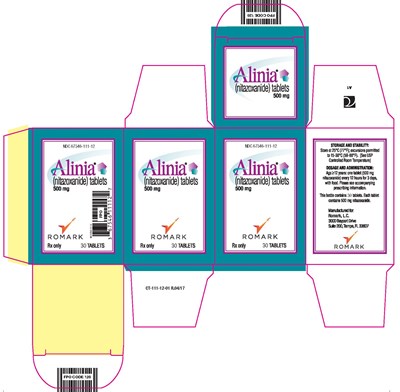
Alinia tablets contain nitazoxanide and are used for the treatment of certain parasitic and viral infections in adults and children over 12 years old.The tablets come in a bottle of 30 and each tablet contains 500mg of the active ingredient nitazoxanide. The tablets should be stored at controlled room temperature between 15-30°C. This medication is manufactured by Romark L.C. This information is in accordance with the codification on the bottle - CT-111-12-01 R0O4A7.*
Alinia Tablets - 12 ct Carton - CT 111 14 02 FINAL PROOF Alina 500mg 12count Carton Rev0
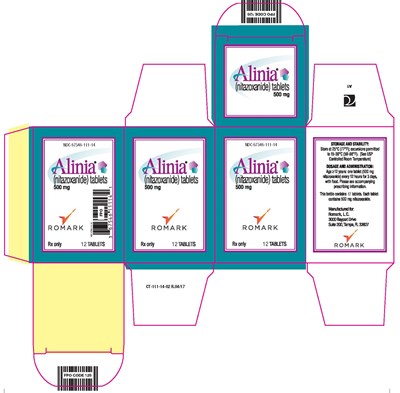
This is a description of a medication called Atinice, containing nitazoxanide tablets of 500mg each. The medication is only available on prescription and is manufactured by Romark, L.C., located in Tampa, AL. The recommended dosage is one tablet (500mg) every 12 hours for three days, with food, for those aged 2-12 years. The medication bottle contains 12 tablets and must be stored at 25°C (77°F) with permissible excursions up to 30°C (86°F).*
Alinia Tablets - 2 ct carton - CT 111 17 01 FINAL PROOF Alina Tablets 2 ct Carton

This text is about a medication called Alinisie (nitazoxanide) that comes in tablets of 500mg. The medication should be stored at temperatures between 15-30°C (59-86°F). Each bottle contains two tablets and the medication is manufactured by Romark, LC. There is also a lot number and expiration date provided.*
Alinia OS Carton - CT 212 21 02 Alina OS Carton Rev 12 18

Alinia is a medication containing nitazoxanide in the form of an oral suspension. It is often prescribed for children, with specific dosages recommended for different age groups. The medication comes in a 60 mL bottle, which must be reconstituted with 48 mL of water and shaken before use. The suspension should be taken with food and any unused portion should be discarded within 7 days of mixing. The medication should be stored at controlled room temperature and must be tightly closed when not in use. The medication is manufactured by Romark and contains 1.2 g of nitazoxanide.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.




