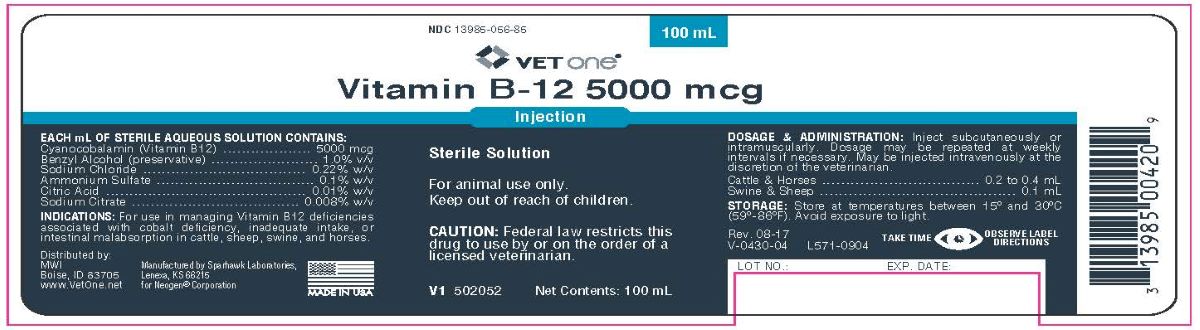
Animal NDC 13985-056-85 Vitamin B-12 5000 Mcg
Cyanocobalamin
Animal Product Information
| Field Name | Field Value |
|---|---|
| Animal NDC Code | 13985-056-85 |
| Proprietary Name | Vitamin B-12 5000 Mcg What is the Proprietary Name? The proprietary name also known as the trade name is the name of the product chosen by the medication labeler for marketing purposes. |
| Non-Proprietary Name | Cyanocobalamin What is the Non-Proprietary Name? The non-proprietary name is sometimes called the generic name. The generic name usually includes the active ingredient(s) of the product. |
| Labeler Name | Mwi/vet One |
| Product Type | Prescription Animal Drug |
| Usage Information |
|
| Active Ingredient(s) |
|
| Inactive Ingredient(s) |
|
| Marketing Category | UNAPPROVED DRUG OTHER What is the Marketing Category? Product types are broken down into several potential Marketing Categories, such as NDA/ANDA/BLA, OTC Monograph, or Unapproved Drug. One and only one Marketing Category may be chosen for a product, not all marketing categories are available to all product types. Currently, only final marketed product categories are included. The complete list of codes and translations can be found at www.fda.gov/edrls under Structured Product Labeling Resources. |
Vitamin B-12 5000 Mcg Images
Vitamin B-12 5000 Mcg Animal Product Labeling Information
The product labeling information includes all published material associated to a drug. Product labeling documents include information like generic names, active ingredients, ingredient strength dosage, routes of administration, appearance, usage, warnings, inactive ingredients, etc.
Table of Contents
Indications:
For use in managing Vitamin B12 deficiencies associated with cobalt deficiency, inadequate intake, or intestinal malabsorption in
cattle, sheep, swine, and horses.
Each Ml Of Sterile Aqueous Solution Contains:
Cyanocobalamin (Vitamin B12)..............5000 mcg
Benzyl Alcohol (preservative).................1.0% v/v
Sodium Chloride.................................0.22% w/v
Ammonium Sulfate...............................0.1% w/v
Citric Acid..........................................0.01% w/v
Sodium Citrate..................................0.008% w/v
Dosage & Administration:
Inject subcutaneously or intramuscularly. Dosage may be repeated at weekly intervals if necessary. May be injected intravenously at the discretion of the veterinarian.
Cattle and Horses ............................... 0.2 to 0.4 mL
Swine and Sheep .......................................... 0.1 mL
Storage:
Store at temperature between 15° and 30°C (59° - 86°F). Avoid exposure to light.
Other
Rev. 08-17
V-0430-04 L571-0904
TAKE TIME OBSERVE LABEL DIRECTIONS
Distributed by:
MWIBoise, ID 83705
www.VetOne.net
Manufactured by Sparhawk Laboratories,
Lenexa, KS 66215
for Neogen® Corporation
MADE IN USA
* The information on this page is for an ANIMAL PRODUCT, please review the complete disclaimer below.