What is NDC?
About NDC Codes
A National Drug Code (NDC) is a unique, standardized numeric identifier assigned to medications and certain healthcare products in the United States. The NDC system was established under the Drug Listing Act of 1972 as part of the Federal Food, Drug, and Cosmetic Act (Section 510, 21 U.S.C. § 360). This system is maintained by the U.S. Food and Drug Administration (FDA) to facilitate drug identification, regulatory compliance, and healthcare operations.
Each NDC consists of a three-segment number that conveys critical information about the product:
- Labeler Code (First Segment) – Identifies the entity responsible for manufacturing, marketing, repackaging, or distributing the drug. The FDA assigns this code.
- Product Code (Second Segment) – Specifies the drug’s formulation, including its strength, dosage form (e.g., tablet, capsule, liquid), and active ingredients. This segment is defined by the labeler.
- Package Code (Third Segment) – Indicates the package size and type, such as a bottle of 30 tablets or a 10 mL vial. This is also determined by the labeler.
NDC Format Example
Let’s break down an example to illustrate how an NDC code is structured:
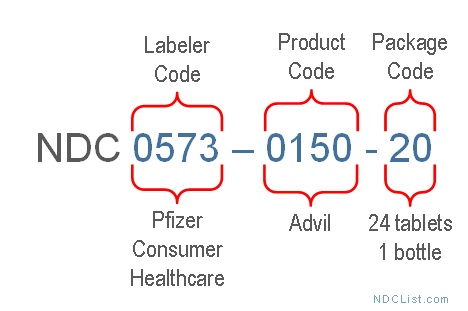
For Advil (Ibuprofen) – 1 bottle with 24 coated tablets, the NDC is 0573-0150-20.
- First Segment (Labeler Code): "0573" – Represents Pfizer Consumer Healthcare, the labeler of Advil.
- Second Segment (Product Code): "0150" – Identifies the specific drug formulation, in this case, Advil (Ibuprofen).
- Third Segment (Package Code): "20" – Denotes the package size, 24 coated tablets in 1 bottle.
Important Note: NDC codes do not indicate FDA approval. Some products listed under an NDC may not be FDA-approved but are still assigned an NDC for tracking purposes.
NDC and Insurance Billing
For insurance reimbursement and electronic transactions, the Centers for Medicare & Medicaid Services (CMS) require an 11-digit NDC format with no hyphens, spaces, or special characters. If an NDC is shorter than 11 digits, it must be padded with leading zeros to maintain the standard 5-4-2 configuration.
Example: Converting an NDC for Billing: The NDC 0573-0150-20 must be formatted as 00573015020 for insurance billing.
| NDC Package Code | NDC Package Format | NDC Billing Code | NDC Billing Format |
|---|---|---|---|
| 0573-0150-20 | XXXX-XXXX-XX | 00573015020 | 0XXXXXXXXXX |
| 66715-9741-3 | XXXXX-XXXX-X | 667150974103 | XXXXXXXXX0X |
| 67751-149-02 | XXXXX-XXX-XX | 67751014902 | XXXXX0XXXXX |
This standardized format ensures seamless communication between pharmacies, insurers, and healthcare providers for claims processing, reimbursement, and inventory management.
Why Are NDC Codes Important?
NDCs are essential in the healthcare, pharmaceutical, and insurance industries for:
- Drug identification and tracking – Ensuring precise identification of medications.
- Pharmacy operations – Facilitating prescription processing and inventory management.
- Insurance claims and reimbursement – Standardizing drug identification for billing purposes.
- Regulatory compliance – Assisting government agencies in monitoring drug distribution.
The NDC system is a fundamental component of drug safety, efficiency, and healthcare coordination in the United States.