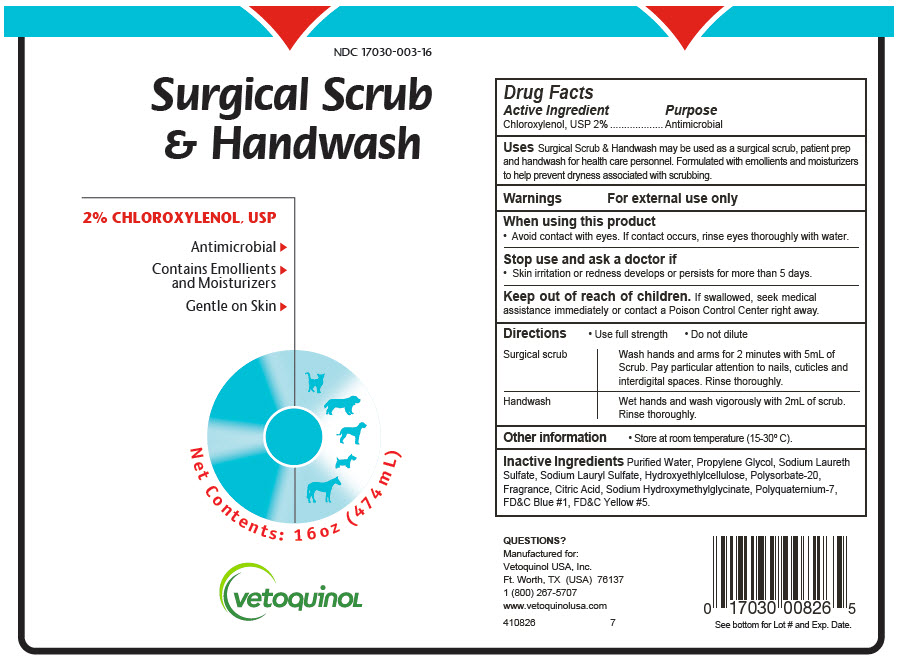
Animal NDC 17030-003-16 Surgical Scrub And Handwash
Chloroxylenol
Animal Product Information
| Field Name | Field Value |
|---|---|
| Animal NDC Code | 17030-003-16 |
| Proprietary Name | Surgical Scrub And Handwash What is the Proprietary Name? The proprietary name also known as the trade name is the name of the product chosen by the medication labeler for marketing purposes. |
| Non-Proprietary Name | Chloroxylenol What is the Non-Proprietary Name? The non-proprietary name is sometimes called the generic name. The generic name usually includes the active ingredient(s) of the product. |
| Labeler Name | Vetoquinol Usa, Inc. |
| Product Type | Otc Animal Drug |
| Usage Information |
|
| Active Ingredient(s) |
|
| Inactive Ingredient(s) |
|
| Marketing Category | UNAPPROVED DRUG OTHER What is the Marketing Category? Product types are broken down into several potential Marketing Categories, such as NDA/ANDA/BLA, OTC Monograph, or Unapproved Drug. One and only one Marketing Category may be chosen for a product, not all marketing categories are available to all product types. Currently, only final marketed product categories are included. The complete list of codes and translations can be found at www.fda.gov/edrls under Structured Product Labeling Resources. |
Surgical Scrub And Handwash Images
Surgical Scrub And Handwash Animal Product Labeling Information
The product labeling information includes all published material associated to a drug. Product labeling documents include information like generic names, active ingredients, ingredient strength dosage, routes of administration, appearance, usage, warnings, inactive ingredients, etc.
Table of Contents
Other
Drug Facts
Active Ingredient
Chloroxylenol, USP 2%
Purpose
Antimicrobial
Keep out of reach of children. If swallowed, seek medical assistance immediately or contact a Poison Control Center right away.
Inactive Ingredients Purified Water, Propylene Glycol, Sodium Laureth Sulfate, Sodium Lauryl Sulfate, Hydroxyethlylcellulose, Polysorbate-20, Fragrance, Citric Acid, Sodium Hydroxymethylglycinate, Polyquaternium-7, FD&C Blue #1, FD&C Yellow #5.
Indications & Usage
Uses Surgical Scrub & Handwash may be used as a surgical scrub, patient prep and handwash for health care personnel. Formulated with emollients and moisturizers to help prevent dryness associated with scrubbing.
Warnings
For external use only
When Using This Product
- Avoid contact with eyes. If contact occurs, rinse eyes thoroughly with water.
Stop Use And Ask A Doctor If
- Skin irritation or redness develops or persists for more than 5 days.
Directions
- Use full strength
- Do not dilute
| Surgical scrub | Wash hands and arms for 2 minutes with 5mL of Scrub. Pay particular attention to nails, cuticles and interdigital spaces. Rinse thoroughly. |
| Handwash | Wet hands and wash vigorously with 2mL of scrub. Rinse thoroughly. |
Other Information
- Store at room temperature (15-30° C).
Questions?
1 (800) 267-5707
Principal Display Panel - 474 Ml Bottle Label
NDC 17030-003-16
Surgical Scrub
& Handwash
2% CHLOROXYLENOL, USP
Antimicrobial ▶
Contains Emollients ▶
and Moisturizers
Gentle on Skin ▶
Net Contents: 16oz (474 mL)
vetoquinoL
* The information on this page is for an ANIMAL PRODUCT, please review the complete disclaimer below.