Animal NDC 17030-100-60 Butequine
Phenylbutazone
Animal Product Information
Butequine Animal Product Labeling Information
The product labeling information includes all published material associated to a drug. Product labeling documents include information like generic names, active ingredients, ingredient strength dosage, routes of administration, appearance, usage, warnings, inactive ingredients, etc.
Table of Contents
Other
NDC 17030 100 60
Approved by FDA under ANADA # 200-266
For Veterinary Use Only
KEEP OUT OF REACH OF CHILDREN
Keep Butequine™ in a secure location out of reach of dogs, cats, and other animals to prevent accidental ingestion or overdose.
CAUTION: Federal (U.S.A.) law restricts this drug to use by or on the order of a licensed veterinarian.
Manufactured for:
Vetoquinol USA, Inc.
Ft. Worth, TX (USA) 76137
www.vetoquinolusa.com
439185
Rev 01/2023
Hold for Canada
Reference #####
Description
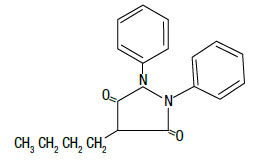
DESCRIPTION: Butequine™ paste is a synthetic, non-hormonal anti-inflammatory, antipyretic compound useful in the management of inflammatory conditions. The apparent analgesic effect is probably related mainly to the compound's anti-inflammatory properties. Chemically, phenylbutazone is 4-butyl-1, 2-diphenyl-3, 5-pyrazolidinedione. It is a pyrazalone derivative, entirely unrelated to the steroid hormones, and has the following structural formula:
Veterinary Indications
INDICATIONS: For the relief of inflammatory conditions associated with the musculoskeletal system in horses.
Contraindications
CONTRAINDICATIONS: Use with caution in patients who have a history of drug allergy.
Warnings
WARNING: Do not use in horses intended for human consumption.
Precautions
PRECAUTIONS: Stop medication at the first sign of gastrointestinal upset, jaundice, or blood dyscrasia. Authenticated cases of agranulocytosis associated with the drug have occurred in man; fatal reactions, although rare, have been reported in dogs after long-term therapy. To guard against the possibility, conduct routine blood counts at weekly intervals during the early phase of therapy and at intervals of two weeks thereafter. Any significant fall in the total white blood cell count, relative decrease in granulocytes, or black or tarry stools should be regarded as a signal for immediate cessation of therapy and institution of appropriate counter-measures. In the treatment of inflammatory conditions associated with infections, specific anti-infective therapy is required.
Dosage & Administration
ADMINISTRATION AND DOSAGE: Orally – 1 to 2 g of phenylbutazone per 500 lb. (227 kg) of body weight daily. Do not exceed 4 g daily. Use a relatively high dose for the first 48 hours, then reduce gradually to a maintenance dose. Maintain lowest dose capable of producing desired clinical response.
Guidelines To Successful Therapy:
- Important - To avoid overdosing and to ensure the correct first dose, move ring on plunger to the appropriate dosage position before administration (see Figure 1). The edge of the ring closest to the syringe barrel should be aligned to the gram/dosage mark on the plunger.
- When administering Butequine™ paste the oral cavity should be empty. Set ring on plunger to deliver the appropriate dosage based on weight (see Figure 1). Deposit paste on the back of tongue by depressing plunger.
- Use a relatively high dose for the first 48 hours (not to exceed 4 g daily), then reduce gradually to a maintenance dose. Maintain lowest dose capable of producing desired clinical response.
- Response to Butequine™ paste therapy is prompt, usually occuring within 24 hours. If no significant clinical response is evident after 5 days, reevaluate diagnosis and therapeutic approach.
- Many chronic conditions will respond to Butequine™ paste therapy, but discontinuance of treatment may result in recurrence of symptoms.
Storage And Handling
STORAGE: Store at 15°-25°C (59°-77°F).
How Supplied
HOW SUPPLIED: Syringes containing 20 g phenylbutazone.
Contact Information:
To report suspected adverse events, contact Vetoquinol USA, Inc. at 1-800-835-9496. For technical assistance or to obtain a copy of the Safety Data Sheet (SDS), contact Vetoquinol USA, Inc. at 1-800-267-5707. For additional information about adverse drug experience reporting for animal drugs, contact FDA at 1-888-FDAVETS or online at www.fda.gov/reportanimalae
Principal Display Panel - 60 Ml Syringe Carton
FOR VETERINARY USE ONLY
Butequine™
(phenylbutazone)
Paste for Horses
NDC 17030 100 60
Approved by FDA under ANADA # 200-266
vetoquinol
Each syringe contains 20 g phenylbutazone
Each 3 mL marking on the plunger contains: phenylbutazone: 1 g
Net Volume: 60 mL
* The information on this page is for an ANIMAL PRODUCT, please review the complete disclaimer below.