Animal NDC 17033-122-75 Animax
Nystatin-neomycin Sulfate-thiostrepton-triamcinolone Acetonide
Animal Product Information
Animax Images
Animax Animal Product Labeling Information
The product labeling information includes all published material associated to a drug. Product labeling documents include information like generic names, active ingredients, ingredient strength dosage, routes of administration, appearance, usage, warnings, inactive ingredients, etc.
Table of Contents
Other
VETERINARY
For use in dogs and cats only.
CAUTION: Federal law restricts this drug to use by or on the order of a licensed veterinarian.
Description:
Animax Ointment combines nystatin, neomycin sulfate, thiostrepton and triamcinolone acetonide in a non-irritating polyethylene and mineral oil base.
nystatin 100,000 units
neomycin sulfate equivalent to neomycin base 2.5 mg
thiostrepton 2,500 units
triamcinolone acetonide 1 mg
The preparation is intended for local therapy in a variety of cutaneous disorders of dogs and cats; it is especially useful in disorders caused, complicated, or threatened by bacterial and/or candidal (monilial) infection.
Actions:
Warnings:
Clinical and experimental data have demonstrated that corticosteroids administered orally or by injection to animals may induce the first stage of parturition if used during the last trimester of pregnancy and may precipitate premature parturition followed by dystocia, fetal death, retained placenta, and metritis.
Additionally, corticosteroids administered to dogs, rabbits and rodents during pregnancy have resulted in cleft palate in the offspring. In dogs, other congenital anomalies have resulted: deformed forelegs, phocomelia, and anasarca.
Indications:
Animax Ointment is particularly useful in the treatment of acute and chronic otitis of varied etiologies, in interdigital cysts in cats and dogs and in anal gland infections in dogs.
The preparation is also indicated in the management of dermatologic disorders characterized by inflammation and dry or exudative dermatitis, particularly those caused, complicated, or threatened by bacterial or candidal (Candida albicans) infections. It is also of value in eczematous dermatitis, contact dermatitis, and seborrheic dermatitis; and as an adjunct in the treatment of dermatitis due to parasitic infestation.
Precautions:
Animax Ointment is not intended for the treatment of deep abscesses or deep-seated infections such as inflammation of the lymphatic vessels. Parenteral antibiotic therapy is indicated in these infections.
Animax Ointment has been extremely well tolerated. Cutaneous reactions attributable to its use have been extremely rare. The occurrence of systemic reactions is rarely a problem with topical administration. There is some evidence that corticosteroids can be absorbed after topical application and cause systemic effects. Therefore, an animal receiving Animax Ointment therapy should be observed closely for signs such as polydipsia, polyuria, and increased weight gain.
Animax Ointment is not generally recommended for the treatment of deep or puncture wounds or serious burns.
Sensitivity to neomycin may occur. If redness, irritation, or swelling persists or increases, discontinue use. Do not use if pus is present since the drug may allow the infection to spread.
Avoid ingestion. Oral or parenteral use of corticosteroids (depending on dose, duration of use, and specific steroid) may result in inhibition of endogenous steroid production following drug withdrawal.
Side Effects:
SAP and SGPT (ALT) enzyme elevations, polydipsia and polyuria, vomiting, and diarrhea (occasionally bloody) have been observed following parenteral or systemic use of synthetic corticosteroids in dogs.
Cushing's syndrome has been reported in association with prolonged or repeated steroid therapy in dogs. Temporary hearing loss has been reported in conjunction with treatment of otitis with products containing corticosteroids. However, regression usually occurred following withdrawal of the drug. Hearing loss, with varying degrees of recovery, has been reported with the use of Animax Ointment. If hearing dysfunction is noted during the course of treatment with Animax Ointment, discontinue its use.
To report suspected adverse drug events, for technical assistance or to obtain a copy of the safety data sheet (SDS), contact Dechra at (866) 933-2472.
For additional information about adverse drug experience reporting for animal drugs, contact FDA at 1-888-FDA-VETS or online at http://www.fda.gov/reportanimalae.
Caution:
Dosage And Administration:
Otitis
Infected Anal Glands, Cystic Areas, etc.
Other Dermatologic Disorders
How Supplied:
7.5 mL tubes NDC 17033-122-75
15 mL tubes NDC 17033-122-15
30 mL tubes NDC 17033-122-30
STORAGE: Store at room temperature; avoid excessive heat (104°F).
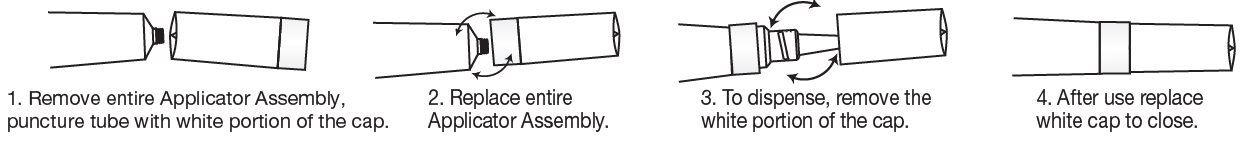
To Open:
Use cap to puncture seal.
Important:
The opening of this product is covered by a metal tamper-evident seal.
If this seal has been punctured or is not visible, do not use and return product to place of purchase.
Approved by FDA under NADA # 140-847
MANUFACTURED FOR:
Dechra Veterinary Products
Overland Park, KS 66211 USA
MANUFACTURED BY:
Fougera Pharmaceuticals Inc.
Melville, NY 11747 USA
Principal Display Panel - 7.5 Ml Carton
Animax® Ointment
(nystatin-neomycin sulfate-thiostrepton-triamcinolone acetonide ointment)
NET CONTENTS 7.5 mL
NDC 17033-122-75
* The information on this page is for an ANIMAL PRODUCT, please review the complete disclaimer below.