Animal NDC 23243-9704-1 Corid Type A Medicated Article
Amprolium
Animal Product Information
Corid Type A Medicated Article Images
Corid Type A Medicated Article Animal Product Labeling Information
The product labeling information includes all published material associated to a drug. Product labeling documents include information like generic names, active ingredients, ingredient strength dosage, routes of administration, appearance, usage, warnings, inactive ingredients, etc.
Table of Contents
Other
CORID® 25%
(amprolium)
Type A Medicated Article
Coccidiostat
Approved by FDA under NADA # 012-350
®CORID is a trademark ot Huvepharma EOOD.
Net Wt.: 50 lb (22.68 kg)
CORID® 25%
(amprolium)
Type A Medicated Article
Distributed by:
Huvepharma, lnc.
Peachtree City, GA 30269
Veterinary Indications
To be used in the preparation of medicated feed to aid
in the prevention and treatment of coccidiosis in calves.
Otc - Active Ingredient
ACTIVE DRUG INGREDIENT: Amprolium ..................... 25%
Inactive Ingredient
INGREDIENTS: Corn Gluten Feed and Soybean Oil
Storage And Handling
SEE DIRECTIONS ON BACK PANEL
STORAGE: Store at or below 77°F (25°C), excursions permitted to 104°F (40°C).
Avoid prolonged exposure to high humidity.
Indications & Usage
INDICATIONS FOR USE:
An aid in the prevention and treatment of coccidiosis caused by Eimeria bovis and E. zurnii in calves.
Residue Warning
WITHDRAWAL PERIODS AND RESIDUE WARNINGS:
Withdraw 24 hours before slaughter. A withdrawal period has not been established for this
product in pre-ruminating calves. Do not use in calves to be processed for veal.
User Safety Warnings
USER SAFETY WARNING:
Not for human use. Keep out of reach of children.
Contact Huvepharma Inc. at 1-877-994-4883 or http://www.huvepharma.us. For additional information about
reporting side effects for animal drugs, contact FDA at 1-888-FDA-VETS or http://www.fda.gov/reportanimalae.
Precautions
CAUTION: For satisfactory diagnosis, a microscopic examination of the feces should be done by a veterinarian or
diagnostic laboratory before treatment. When treating outbreaks, drug should be administered promptly after
diagnosis is determined.
Do not use in feeds containing bentonite.
Use as the sole source of amprolium.
Restricted Drug (California) – Use only as directed.
Dosage & Administration
MIXING DIRECTIONS: CORID 25% (amprolium) Type A medicated article should be thoroughly and evenly mixed
in the feed. CORID 25% may be used to manufacture a Type B medicated feed with a concentration of 2.5%
amprolium or a Type C medicated feed in the concentration range of 0.0125% to 1.25% amprolium.
• Use 200 pounds of CORID 25% per ton of feed to produce
Type B medicated feed containing 2.5% amprolium.
• Use 1 pound of CORID 25% per ton of feed to produce
Type C medicated feed containing 0.0125% amprolium.*
• Use 100 pounds of CORID 25% per ton of feed to produce
Type C medicated feed containing 1.25% amprolium.
• Use 10 pounds of 2.5% Type B per ton of feed to produce
Type C medicated feed containing 0.0125% amprolium.
• Use 1000 pounds of 2.5% Type B per ton of feed to produce
Type C medicated feed containing 1.25% amprolium.
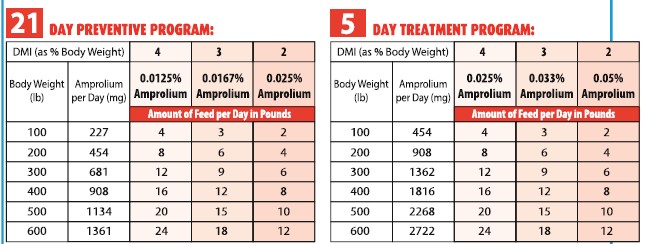
AMOUNT AND DIRECTIONS FOR USE:
Prevention: 227 mg amprolium /100 lb (5 mg/kg) body weight per day for 21 days during periods of exposure or when
experience indicates that coccidiosis is likely to be a hazard.
Treatment: 454 mg amprolium/100 lb (10 mg/kg) bodyweight per day for 5 days. Use on a herd basis only; when one
or more calves show signs of coccidiosis, it is likely that the rest of the group has been exposed and all
calves in the group should be treated.
*To aid in even distribution of CORID 25% in the finished feed, prepare a mixture of CORID 25% in a portion of complete feed
ingredient before mixing into the finished ration. Blend the mixture with the remainder of the finished feed and mix thoroughly.
The proper amount of amprolium supplement is mixed with the amount of ration consumed in one day. The tables below
give suggested examples for using Type C medicated feed.
* The information on this page is for an ANIMAL PRODUCT, please review the complete disclaimer below.