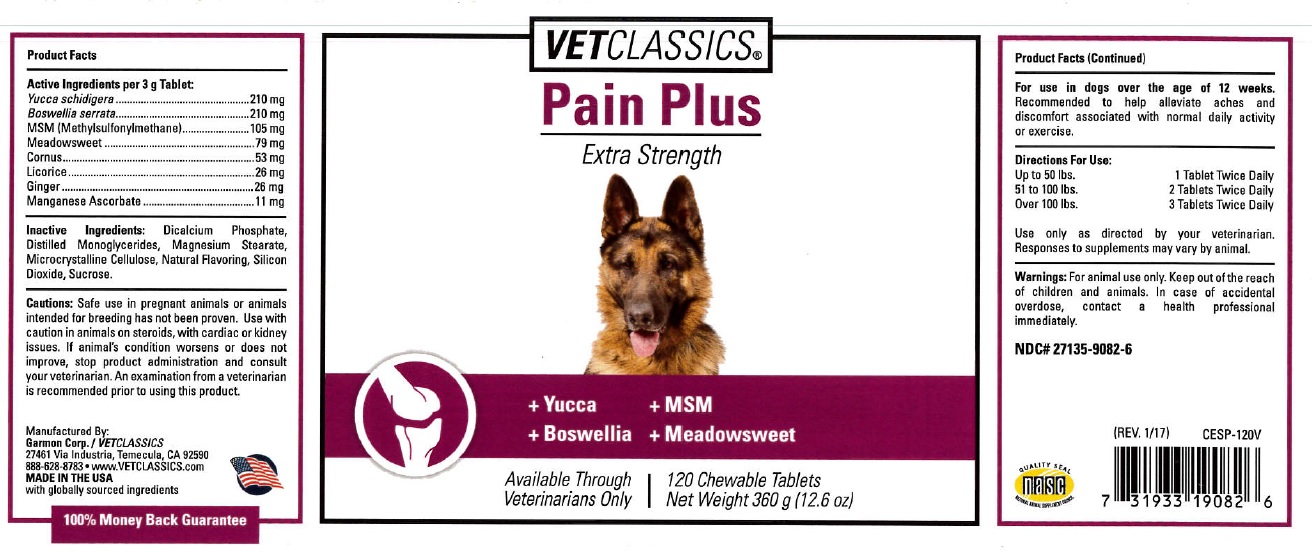
Animal NDC 27135-9082-6 Pain Plus Tab
Animal Product Information
| Field Name | Field Value |
|---|---|
| Animal NDC Code | 27135-9082-6 |
| Proprietary Name | Pain Plus Tab What is the Proprietary Name? The proprietary name also known as the trade name is the name of the product chosen by the medication labeler for marketing purposes. |
| Non-Proprietary Name | Pain Plus Tab What is the Non-Proprietary Name? The non-proprietary name is sometimes called the generic name. The generic name usually includes the active ingredient(s) of the product. |
| Labeler Name | Garmon Corp |
| Product Type | Otc Animal Drug |
| Active Ingredient(s) |
|
| Inactive Ingredient(s) |
|
| Marketing Category | UNAPPROVED DRUG OTHER What is the Marketing Category? Product types are broken down into several potential Marketing Categories, such as NDA/ANDA/BLA, OTC Monograph, or Unapproved Drug. One and only one Marketing Category may be chosen for a product, not all marketing categories are available to all product types. Currently, only final marketed product categories are included. The complete list of codes and translations can be found at www.fda.gov/edrls under Structured Product Labeling Resources. |
Pain Plus Tab Images
Pain Plus Tab Animal Product Labeling Information
The product labeling information includes all published material associated to a drug. Product labeling documents include information like generic names, active ingredients, ingredient strength dosage, routes of administration, appearance, usage, warnings, inactive ingredients, etc.
Table of Contents
Product Facts
Active Ingredients per 3 g Tablet:
Yucca schidigera 210 mg
Boswellia serrata 210 mg
MSM (methylsulfonylmethane) 105 mg
Meadowsweet 79 mg
Cornus 53 mg
Licorice 26 mg
Ginger 26 mg
Manganese Ascorbate 11 mg
Inactive Ingredient
Inactive Ingrdients: Dicalcium Phosphate, Distilled Monoglycerides, Manganesium Stearate, Microcrystalline Cellulose, Natural flavoring, Silicon Dioxide, Sucrose.
Warnings And Precautions
Cautions: Safe use in pregnant animals or animals intended for breeding has not been proven. Use with caution in animals on steriods, with cardiac or kidney issues. If animal's condition worsens or does not improve, stop product administration and consult your veterinarian. An examination from a veterinarian is recommended prior to using this product.
Product Facts (Continued)
For use in dogs over the age of 12 weeks. Recommended to help alleviate aches and discomfort associated with normal daily activity or excercise.
Dosage & Administration
Directions For Use:
Up to 50 lbs. 1 Tablet Twice Daily
51 to 100 lbs. 2 Tablets Twice Daily
Over 100 lbs. 3 TabletsTwice Daily
Use only as directed by your veterinarian. Responses to supplements may vary by animal.
General Precautions
Warnings: For animal use only. Keep out of the reach of children and animals. In case of accidental overdose, contact a health professional immediately.
* The information on this page is for an ANIMAL PRODUCT, please review the complete disclaimer below.