Animal NDC 50989-344-15 Emulsivit E Ad
D-alpha Tocopherol Vitamin A Vitamin D3
Animal Product Information
| Field Name | Field Value |
|---|---|
| Animal NDC Code | 50989-344-15 |
| Proprietary Name | Emulsivit E Ad What is the Proprietary Name? The proprietary name also known as the trade name is the name of the product chosen by the medication labeler for marketing purposes. |
| Non-Proprietary Name | D-alpha Tocopherol Vitamin A Vitamin D3 What is the Non-Proprietary Name? The non-proprietary name is sometimes called the generic name. The generic name usually includes the active ingredient(s) of the product. |
| Labeler Name | Vedco, Inc. |
| Product Type | Otc Animal Drug |
| Usage Information |
|
| Active Ingredient(s) |
|
| Marketing Category | UNAPPROVED DRUG OTHER What is the Marketing Category? Product types are broken down into several potential Marketing Categories, such as NDA/ANDA/BLA, OTC Monograph, or Unapproved Drug. One and only one Marketing Category may be chosen for a product, not all marketing categories are available to all product types. Currently, only final marketed product categories are included. The complete list of codes and translations can be found at www.fda.gov/edrls under Structured Product Labeling Resources. |
Emulsivit E Ad Images
Emulsivit E Ad Animal Product Labeling Information
The product labeling information includes all published material associated to a drug. Product labeling documents include information like generic names, active ingredients, ingredient strength dosage, routes of administration, appearance, usage, warnings, inactive ingredients, etc.
Table of Contents
Indications & Usage
EmulsiVit E/A&D
Injectable Tocopherol/AD
Indications: EmulsiVitE/A&D Injectable d-alpha tocopherol with AD is a clear, sterile, non-aqueous solution of vitamin A,vitamin D3,and vitamin E. The product is intended as a supplemental source of vitamins A, D and E.
Natural vitamin A (carotenes), and tocopherols can be destroyed in feedstuffs through processing, ensiling and storage. Due to these losses, reduced intakes of fat-soluble vitamins can occur in animals maintained in continual confinement compared to animals allowed to graze in lush pasture. Intramuscular or subcutaneous injections offer an efficient and rapid method to increase vitamin A, vitamin D and Vitamin E status of animals.
Each Ml Contains:
Active Ingredients:
vitamin E (as d-alpha-tocopherol,
a natural source of vitamin E).....................................300 IU
Vitamin A Propionate...........................................100,000 IU
Vitamin D3...........................................................10,000 IU
Inactive Ingredients: 2% benzyl alcohol (preservative)
compounder in a water emulsifiable base.
Caution:
Anaphylactoid or allergic reactions, which have resulted in deaths, have been reported in individual animals as well as entire herds. Should such reactions or hypersensitivity occur, treat immediately with injection of epinephrine and/or antihistamines. Do not exceed recomended dosage. Administration of this product to well-nourished animals may cause hypervitaminosis D, which may result in hypercalcemia and hyperphosphatemia.
Warning:
Do not add water to solution.
Not for human use.
Storage And Handling
Store between 15°—30°C (59°—86°F) in a dark place
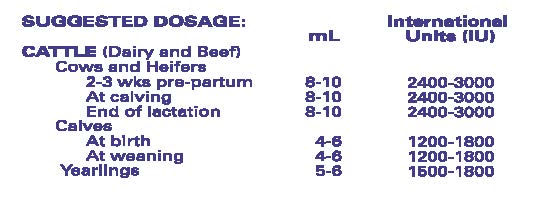
Administration And Dosage:
Intramuscular or Subcutaneous Administration Only. May be repeated. If dose is greater than 5 mL, equally divide the dose and inject at two sites.
* The information on this page is for an ANIMAL PRODUCT, please review the complete disclaimer below.