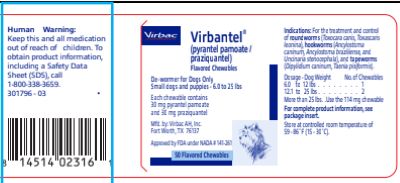
Animal NDC 51311-738-10 Virbantel
Pyrantel Pamoate/praziquantel
Animal Product Information
Virbantel Images
Virbantel Animal Product Labeling Information
The product labeling information includes all published material associated to a drug. Product labeling documents include information like generic names, active ingredients, ingredient strength dosage, routes of administration, appearance, usage, warnings, inactive ingredients, etc.
Table of Contents
Package Contents:
Bottle of 50 flavored chewables
Drug Facts
Active Ingredients (in each chewable): pyrantel pamoate (30 mg) and praziquantel (30 mg)
Purpose: De-wormer for Small Dogs and Puppies Only (6.0 to 25 pounds).
Uses: For The Treatment And Control Of:
- Roundworms (Toxocara canis, Toxascaris leonina)
- Hookworms (Ancylostoma caninum, Ancylostoma braziliense, and Uncinaria stenocephala)
- Tapeworms (Dipylidium caninum, Taenia pisiformis)
Human Warning:
Keep this and all medication out of the reach of children. To obtain product information, including a Safety Data Sheet (SDS),
call 1-800-338-3659.
When Using This Product:
• Consult your veterinarian for assistance in the diagnosis, treatment, and control of parasitism.
• Do not de-worm a dog or puppy that is sick. Consult a veterinarian for diagnosis of the illness.
• VIRBANTEL Flavored Chewables are safe for use in puppies 12 weeks or older and adult dogs. Safety in breeding dogs and pregnant bitches
has not been tested.
You May Notice:
Vomiting, loose stools (with or without blood) and decreased activity following treatment. If you notice these signs, contact a veterinarian.
Directions:
Each flavored chewable contains 30 mg of pyrantel pamoate and 30 mg of praziquantel. The dose for each drug is 2.27 mg per pound of body weight (5 mg/kg). Please refer to the following dosing table for help finding the right dose for your dog.
VIRBANTEL® Flavored Chewables Dosing Table
- You should weigh your dog to make sure you are giving the right dose.
- Virbantel Flavored Chewables are palatable if offered by hand. If your dog does not voluntarily eat the chewable, you can hide the chewable in a small amount of food or place it in the back of the dog's mouth for forced swallowing.
- Make sure that the dog eats the complete dose.
- Watch your dog for a few minutes after dosing to make sure the chewable is not rejected.
Other Information
Recommended De-Worming Schedule:Consult your veterinarian for assistance in the diagnosis, treatment, and control of parasitism. De-worming schedules may vary depending on the climate where you live and the activity of your dog.
Re-treatment: Re-treatment of your dog may be necessary as determined by laboratory fecal examination and/or if your dog is living where re-infections are likely to occur. Consult your veterinarian for assistance in the diagnosis and prevention of re-infection. In case of re-infection with tapeworms (Dipylidium caninum), consult your veterinarian for advice on how to remove fleas from the dog and the environment.
Manufactured By:
Virbac AH, Inc.
Fort Worth, TX 76137
Storage:
Store at controlled room temperature of 59-86˚F (15-30˚C).
Questions? Comments?
To report a suspected adverse reaction, call 1-800-338-3659.
02/19 301798 - 03
Approved by FDA under NADA # 141-261
©2019 Virbac Corporation.
All Rights Reserved.
VIRBANTEL is a registered
trademark of Virbac S.A.
* The information on this page is for an ANIMAL PRODUCT, please review the complete disclaimer below.