Animal NDC 52003-916-20 Nitrogen
Animal Product Information
Nitrogen Images
Nitrogen Animal Product Labeling Information
The product labeling information includes all published material associated to a drug. Product labeling documents include information like generic names, active ingredients, ingredient strength dosage, routes of administration, appearance, usage, warnings, inactive ingredients, etc.
Table of Contents
Warnings And Precautions Section
Should the material not be delivered to a NF customer, the material is “Not Approved for Human Drug Use.”
Package Label.Principal Display Panel
Messer
Gases for Life
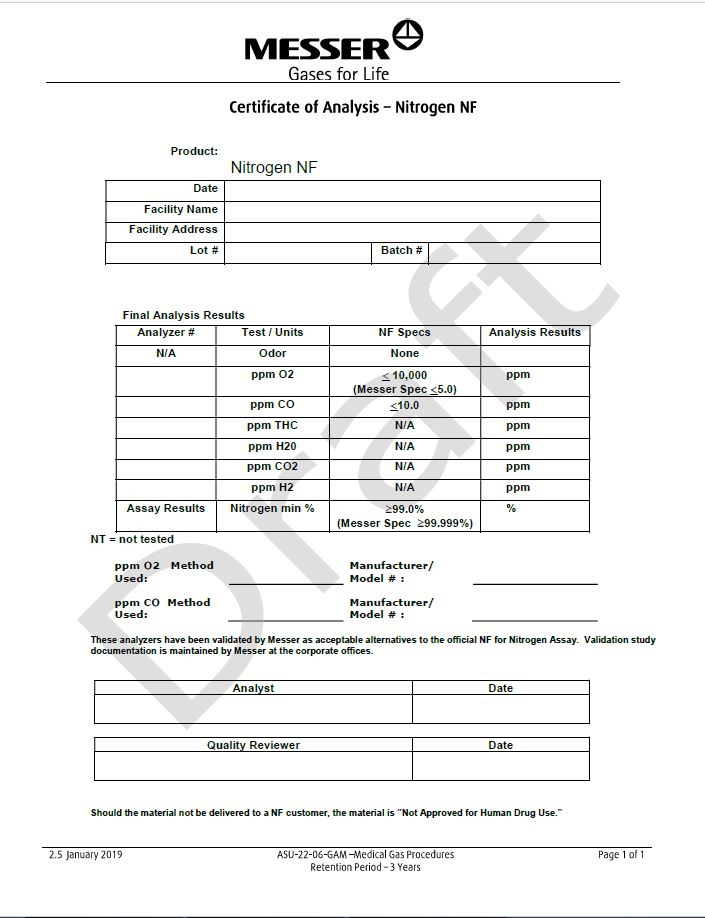
Certicate of Analysis - Nitrogen NF
Product: Nitrogen NF
Date |
| ||
Facility Name |
| ||
Facility Address |
| ||
Lot# |
| Batch # |
|
Final Analysis Results
Analyzer # | Test/Units | NF Specs | Analysis Results |
N/A | Odor | None |
|
| ppm O2 | < 10,000 (Messer Spec <5.0) | ppm |
| ppm CO | < 10.0 | ppm |
| ppm THC | N/A | ppm |
| ppm H2O | N/A | ppm |
| ppm CO2 | N/A | ppm |
| ppm H2 | N/A | ppm |
Assay Results | Nitrogen min % | > 99.0% (Messer Spec >99.999%) | % |
NT= not tested
PPM O2 Method Used: __________________________ | Manufacturer/Model #: __________________________ |
PPM O2 Method Used: __________________________ | Manufacturer/Model #: __________________________ |
This analyzer has been validated by Messer as acceptable alternatives to the official NF for Nitrogen Assay. Validation study documentation is maintained by Messer at the corporate offices.
Analyst | Date |
|
|
|
|
Quality Reviewer | Date |
|
|
|
|
| ||
Should the material not be delivered to a NF customer, the material is “Not Approved for Human Drug Use.”
2.5 January 2019
ASU-22-06-GAM-Medical Gas Procedures
Page 1 of 1
Retention Period – 3 Years
* The information on this page is for an ANIMAL PRODUCT, please review the complete disclaimer below.