Animal NDC 54771-1003-1 Aureomycin
Chlortetracycline Hydrochloride
Animal Product Information
Aureomycin Images
Aureomycin Animal Product Labeling Information
The product labeling information includes all published material associated to a drug. Product labeling documents include information like generic names, active ingredients, ingredient strength dosage, routes of administration, appearance, usage, warnings, inactive ingredients, etc.
Table of Contents
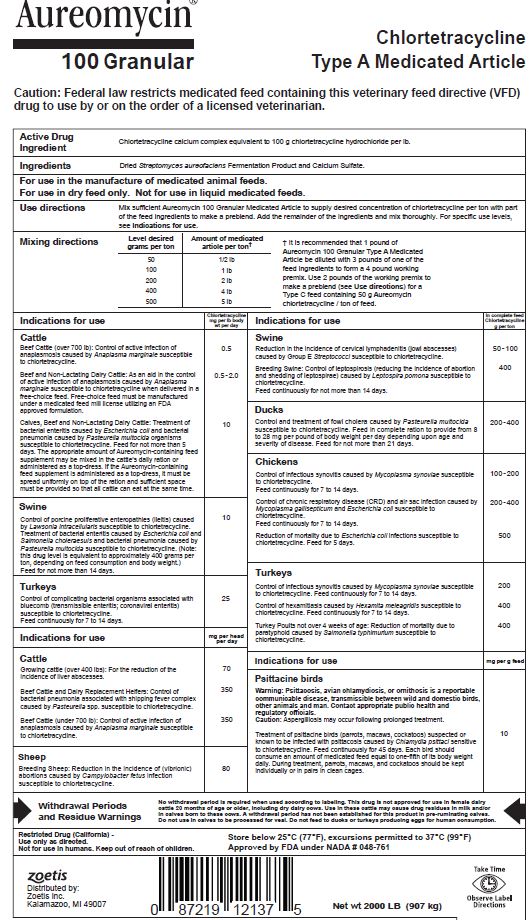
Active Drug Ingredient
Chlortetracycline calcium complex equivalent to 100 g chlortetracycline hydrochloride per lb.
Ingredients
Dried Streptomyces aureofaciens Fermentation Product and Calcium Sulfate.
Use Directions
Mix sufficient Aureomycin 100 Granular Medicated Article to supply desired concentration of chlortetracycline per ton with part of the feed ingredients to make a preblend. Add the remainder of the ingredients and mix thoroughly. For specific use levels, see Indications for use.
Mixing Directions
| Level desired grams per ton | Amount of medicated article per ton* |
|---|---|
50 | 1/2 lb |
100 | 1 lb |
200 | 2 lb |
400 | 4 lb |
500 | 5 lb |
† It is recommended that 1 pound of Aureomycin 100 Granular Type A Medicated Article be diluted with 3 pounds of one of the feed ingredients to form a 4 pound working premix. Use 2 pounds of the working premix to make a preblend (see Use directions) for a Type C feed containing 50 g Aureomycin chlortetracycline / ton of feed.
Indications For Use
| Indications for use | Chlortetracycline mg per lb body wt per day |
| Cattle | |
| Beef Cattle (over 700 lb): Control of active infection of anaplasmosis caused by Anaplasma marginale susceptible to chlortetracycline. | 0.5 |
| Beef and Non-Lactating Dairy Cattle: As an aid in control of active infection of anaplasmosis caused by Anaplasma marginale susceptible to chlortetracycline when delivered in a free-choice feed. Free-choice feed must be manufactured under a medicated feed mill license utilizing an FDA approved formulation. | 0.5-2.0 |
| Calves, Beef and Non-Lactating Dairy Cattle: Treatment of bacterial enteritis caused by Escherichia coli and bacterial pneumonia caused by Pasteurella multocida organisms susceptible to chlortetracycline. Feed for not more than 5 days. The appropriate amount of Aureomycin-containing feed supplement may be mixed in the cattle's daily ration or administered as a top-dress. If the Aureomycin-containing feed supplement is administered as a top-dress, it must be spread uniformly on top of the ration and sufficient space must be provided so that all cattle can eat at the same time. | 10 |
| Swine | |
Control of porcine proliferative enteropathies (ileitis) caused by Lawsonia intracellularis susceptible to chlortetracycline. Feed for not more than 14 days. | 10 |
| Turkeys | |
| Control of complicating bacterial organisms associated with bluecomb (transmissible enteritis; coronaviral enteritis) susceptible to chlortetracycline. Feed continuously for 7 to 14 days. | 25 |
| Indications for use | mg per head per day |
| Cattle | |
| Growing Cattle (over 400 lb): For the reduction of the incidence of liver abscesses. | 70 |
| Beef Cattle and Dairy Replacement Heifers: Control of bacterial pneumonia associated with shipping fever complex caused by Pasteurella spp. susceptible to chlortetracycline. | 350 |
| Beef Cattle (under 700 lb): Control of active infection of anaplasmosis caused by Anaplasma marginale susceptible to chlortetracycline. | 350 |
| Sheep | |
| Breeding Sheep: Reduction in the incidence of (vibrionic) abortions caused by Campylobacter fetus infection susceptible to chlortetracycline. | 80 |
| Indications for use | In complete feed Chlortetracycline g per ton |
| Swine | |
| Reduction in the incidence of cervical lymphadenitis (jowl abscesses) caused by Group E Streptococci susceptible to chlortetracycline. | 50-100 |
| Breeding Swine: Control of leptospirosis (reducing the incidence of abortion and shedding of leptospirae) caused by Leptospira pomona susceptible to chlortetracycline. Feed continuously for not more than 14 days. | 400 |
| Ducks | |
| Control and treatment of fowl cholera caused by Pasteurella multocida susceptible to chlortetracycline. Feed in complete ration to provide from 8 to 28 mg per pound of body weight per day depending upon age and severity of disease. Feed for not more than 21 days. | 200-400 |
| Chickens | |
| Control of infectious synovitis caused by Mycoplasma synoviae susceptible to chlortetracycline. Feed continuously for 7 to 14 days. | 100-200 |
| Control of chronic respiratory disease (CRD) and air sac infection caused by Mycoplasma gallisepticum and Escherichia coli susceptible to chlortetracycline. Feed continuously for 7 to 14 days. | 200-400 |
| Reduction of mortality due to Escherichia coli infections susceptible to chlortetracycline. Feed for 5 days. | 500 |
| Turkeys | |
| Control of infectious synovitis caused by Mycoplasma synoviae susceptible to chlortetracycline. Feed continuously for 7 to 14 days. | 200 |
| Control of hexamitiasis caused by Hexamita meleagridis susceptible to chlortetracycline. Feed continuously for 7 to 14 days. | 400 |
| Turkey Poults not over 4 weeks of age: Reduction of mortality due to paratyphoid caused by Salmonella typhimurium susceptible to chlortetracycline. | 400 |
| Indications for use | mg per g feed |
| Psittacine birds | |
| Warning: Psittacosis, avian chlamydiosis, or ornithosis is a reportable communicable disease, transmissible between wild and domestic birds, other animals and man. Contact appropriate public health and regulatory officials. | |
| Caution: Aspergilliosis may occur following prolonged treatment. | |
| Treatment of psittacine birds (parrots, macaws, cockatoos) suspected or known to be infected with psittacosis caused by Chlamydia psittaci sensitive to chlortetracycline. Feed continuously for 45 days. Each bird should consume an amount of medicated feed equal to one-fifth of its body weight daily. During treatment, parrots, macaws, and cockatoos should be kept individually or in pairs in clean cages. | 10 |
Withdrawal Periods And Residue Warnings
No withdrawal period is required when used according to labeling. This drug is not approved for use in female dairy cattle 20 months of age or older, including dry dairy cows. Use in these cattle may cause drug residues in milk and/or in calves born to these cows. A withdrawal period has not been established for this product in pre-ruminating calves. Do not use in calves to be processed for veal. Do not feed to ducks or turkeys producing eggs for human consumption.
Storage
Store below 25°C (77°F), excursions permitted to 37°C (99°F)
Restricted Drug (California) - Use only as directed. Not for use in humans. Keep out of reach of children.
Approved by FDA under NADA # 048-761
Principal Display Panel - 50 Lb Bag
y
40046195
* The information on this page is for an ANIMAL PRODUCT, please review the complete disclaimer below.