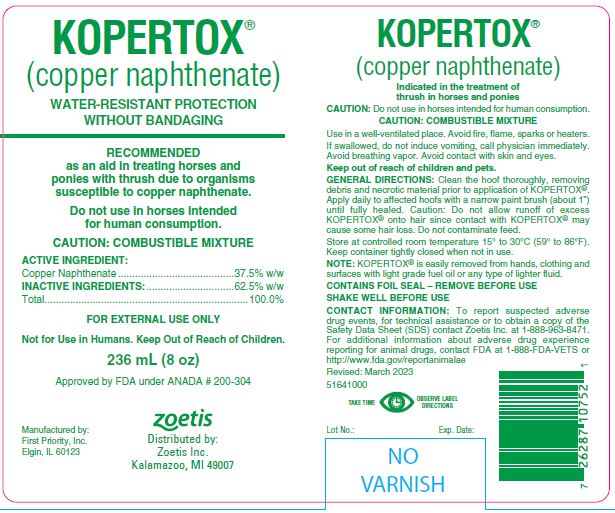
Animal NDC 54771-1100-2 Kopertox
Copper Naphthenate
Animal Product Information
Kopertox Animal Product Labeling Information
The product labeling information includes all published material associated to a drug. Product labeling documents include information like generic names, active ingredients, ingredient strength dosage, routes of administration, appearance, usage, warnings, inactive ingredients, etc.
Table of Contents
Indications & Usage
(copper naphthenate)
WATER-RESISTANT PROTECTION WITHOUT BANDAGING
RECOMMENDED as an aid in treating horses and ponies with thrush due to organisms susceptible to copper naphthenate.
Indicated in the treatment of thrush in horses and ponies
Precautions
Do not use in horses intented for human consumption.
CAUTION: COMBUSTIBLE MIXTURE
FOR EXTERNAL USE ONLY
Not for Use in Humans.
Keep Out of Reach of Children.
Active Ingredient:
Copper Naphthenate ..................... 37.5% w/w
Inactive Ingredients:
........... 62.5% w/w
Total .....................................................100.0%
General Precautions
CAUTION: Do not use in horses intended for human consumption.
CAUTION: COMBUSTIBLE MIXTURE
Use in a well-ventilated place. Avoid fire, flame, sparks or heaters.
If swallowed, do not induce vomiting, call physician immediately. Avoid breathing vapor. Avoid contact with skin and eyes.
Keep out of reach of children and pets.
General Directions:
Clean the hoof thoroughly, removing debris and necrotic material prior to application of KOPERTOX®. Apply daily to affected hoofs with a narrow paint brush (about 1") until fully healed. Caution: Do not allow runoff of excess KOPERTOX® onto hair since contact with KOPERTOX® may cause some hair loss. Do not contaminate feed.
Storage And Handling
Store at controlled room temperature 15° to 30°C (59° to 86°F). Keep container tightly closed when not in use.
Information For Owners/Caregivers
NOTE: KOPERTOX® is easily removed from hands, clothing and surfaces with light grade fuel oil or any type of lighter fluid.
CONTAINS FOIL SEAL – REMOVE BEFORE USE.
SHAKE WELL BEFORE USE.
Contact Information:
To report suspected adverse drug events, for technical assistance or to obtain a copy of the Safety Data Sheet (SDS) contact Zoetis Inc. at
1-888-963-8471. For additional information about adverse drug experience reporting for animal drugs, contact FDA at 1-888-FDA-VETS or http://www.fda.gov/reportanimalae
How Supplied
236 mL (8 oz) Rev. 03/2023 5164100
473 mL (16 oz) Rev. 03/2023 5161100
Approved by FDA under ANADA # 200-304
Distributed by:
Zoetis Inc.
Kalamazoo, MI 49007
Manufactured by:
First Priority, Inc.
Elgin, IL 60123
236 Ml (8 Oz)
* The information on this page is for an ANIMAL PRODUCT, please review the complete disclaimer below.