Animal NDC 54771-3903-2 Synovex C
Progesterone And Estradiol Benzoate
Animal Product Information
Synovex C Animal Product Labeling Information
The product labeling information includes all published material associated to a drug. Product labeling documents include information like generic names, active ingredients, ingredient strength dosage, routes of administration, appearance, usage, warnings, inactive ingredients, etc.
Table of Contents
Withdrawal Periods And Residue Warnings
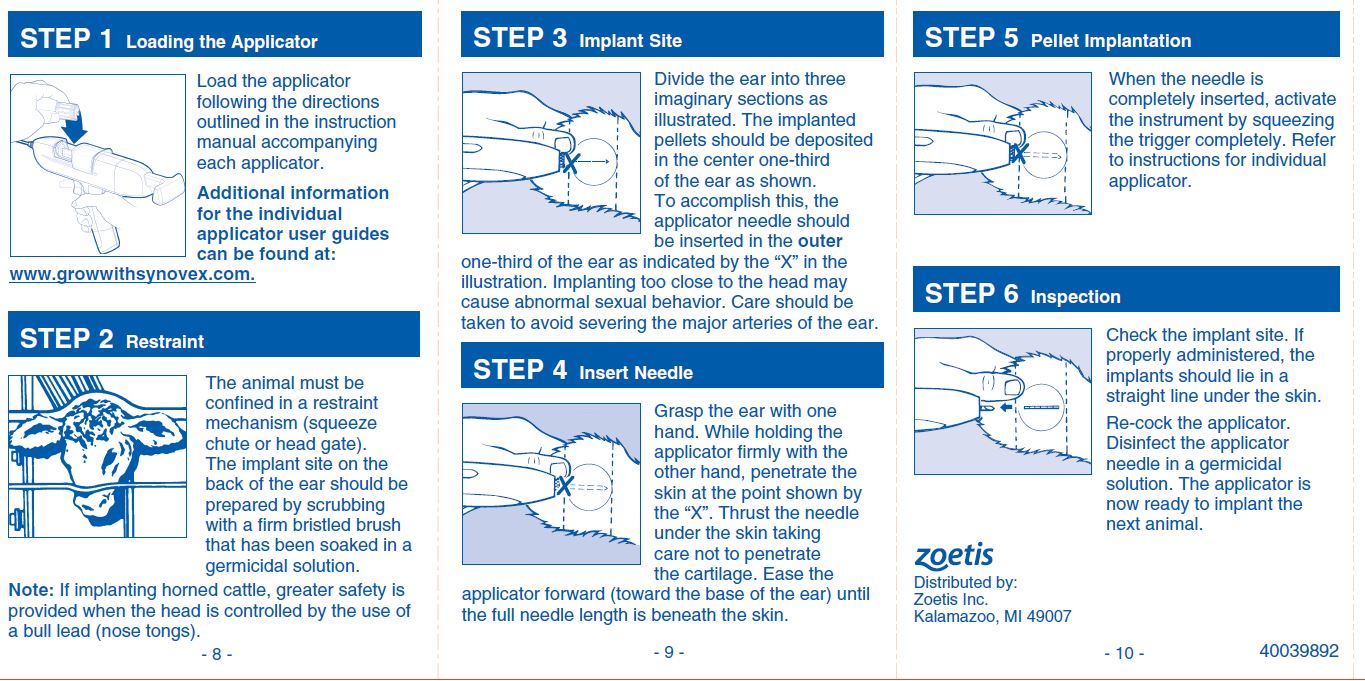
No withdrawal period is required when used according to labeling. Do not use in beef calves less than 45 days of age, dairy calves, and veal calves. Do not use in bull calves intended for reproduction. A withdrawal period has not been established for this product in pre-ruminating calves. Do not use in dairy cows or in animals intended for subsequent breeding. Use in these cattle may cause drug residues in milk and/or in calves born to these cows. Implant pellets subcutaneously in ear only. Any other location is a violation of Federal Law. Do not attempt salvage of implanted site for human or animal food.
User Safety Warnings
Not for use in humans. Keep out of reach of children.
Animal Safety Warnings
Bulling has occasionally been reported in implanted steers and heifers. Vaginal and rectal prolapse, udder development, ventral edema and elevated tailheads have occasionally been reported in heifers administered SYNOVEX® C implants.
Directions
Implant complete contents of one cartridge cell to each steer or heifer at each implanting. Approved implantation technique is fully described in the package insert.
NOTE: Never sacrifice careful, clean technique for speed of implantation.
Indications For Use
• For increased rate of weight gain in beef calves 45 days of age and older and weighing up to 400 lbs.
• For increased rate of weight gain in growing beef steers fed in confinement for slaughter in a reimplantation program where SYNOVEX® C is the first implant and a SYNOVEX® S implant is administered approximately 70 days later.
• Other than as described on the labeling, this implant is not approved for repeated implantation (reimplantation) with any other cattle ear implant as safety and effectiveness have not been evaluated.
Do not use in calves less than 45 days of age or veal calves because effectiveness and safety have not been evaluated.
Do not use in animals intended for subsequent breeding, or in dairy cows.
Storage
Store at controlled room temperature 20° - 25°C (68° - 77°F) with excursions between 15°-30°C (59°-86°F). Avoid excessive heat or humidity.
Questions/Comments?
For a copy of the Safety Data Sheet or to report side effects, contact Zoetis Inc. at 1-888-963-8471. For additional information about reporting side effects for animal drugs, contact FDA at 1-888-FDA-VETS or http://www.fda.gov/reportanimalae.
Principal Display Panel - 10 Cartridge Implant Carton
40039892
* The information on this page is for an ANIMAL PRODUCT, please review the complete disclaimer below.