Animal NDC 54771-9847-1 Advocin
Danofloxacin Mesylate
Animal Product Information
Advocin Animal Product Labeling Information
The product labeling information includes all published material associated to a drug. Product labeling documents include information like generic names, active ingredients, ingredient strength dosage, routes of administration, appearance, usage, warnings, inactive ingredients, etc.
Table of Contents
- Caution
- Description
- Indications
- Dosage And Administration
- Residue Warnings
- Human Warnings
- Precautions
- Adverse Reactions
- (A) Pharmacokinetics
- (B) Microbiology
- Effectiveness
- Animal Safety
- Toxicology
- Storage Information
- How Supplied
- Contact Information
- Principal Display Panel - 100 Ml Bottle Label
- Principal Display Panel - 250 Ml Bottle Label
Caution
Federal law restricts this drug to use by or on the order of a licensed veterinarian. Federal law prohibits the extra-label use of this drug in food-producing animals.
Description
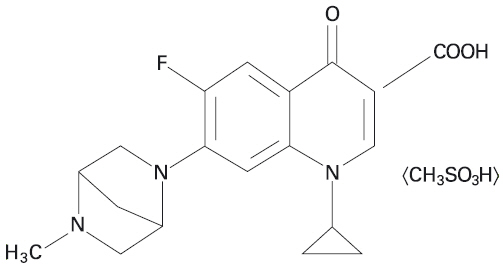
ADVOCIN is a sterile injectable solution containing danofloxacin mesylate, a synthetic fluoroquinolone antimicrobial agent. Danofloxacin mesylate is the non-proprietary designation for (1S)-1cyclopropyl-6-fluoro-1,4-dihydro-7-(5-methyl-2,5-diazabicyclo [2.2.1]hept-2-yl)-4-oxo-3-quinolone carboxylic acid monomethanesulfonate. The empirical formula is C19H20FN3O3 • CH3SO3H and the molecular weight is 453.49.
Figure 1. The chemical structure of danofloxacin mesylate.
Each mL contains 180 mg of danofloxacin as the mesylate salt, 200 mg 2-pyrrolidone, 50 mg polyvinyl pyrrolidone, 20.3 mg heavy magnesium oxide, 2.5 mg phenol, 5 mg monothioglycerol, hydrochloric acid or sodium hydroxide as needed to adjust pH, nitrogen headspace and water for injection, q.s.
Indications
For the treatment of bovine respiratory disease (BRD) associated with Mannheimia haemolytica and Pasteurella multocida in beef cattle and for the control of BRD in beef cattle at high risk of developing BRD associated with Mannheimia haemolytica and Pasteurella multocida.
Dosage And Administration
Care should be taken to dose accurately. Administered dose volume should not exceed 15 mL per injection site.
Single-Dose Therapy (BRD Treatment and Control in Cattle at High Risk):
Administer subcutaneously at 8 mg/kg of body weight (2 mL/100 lb) as a one-time injection.
Multi-Day Therapy (BRD Treatment):
Administer subcutaneously at 6 mg/kg of body weight (1.5 mL/100 lb) with this treatment repeated once approximately 48 hours following the first injection.
| Dose Volume (mL) | ||
|---|---|---|
| Cattle Weight (lb) | 6 mg/kg, given twice, 48 hours apart (treatment) | 8 mg/kg given once (treatment and control in cattle at high risk) |
| 50 | 0.75 | 1 |
| 100 | 1.5 | 2 |
| 150 | 2.25 | 3 |
| 200 | 3 | 4 |
| 250 | 3.75 | 5 |
| 300 | 4.5 | 6 |
| 400 | 6 | 8 |
| 500 | 7.5 | 10 |
| 600 | 9 | 12 |
| 700 | 10.5 | 14 |
| 800 | 12 | 16 Administered dose volume should not exceed 15 mL per injection site. |
| 900 | 13.5 | 18 |
| 1000 | 15 | 20 |
* Administered dose volume should not exceed 15 mL per injection site.
Residue Warnings
Animals intended for human consumption must not be slaughtered within 4 days from the last treatment. Do not use in cattle intended for dairy production. A withdrawal period has not been established for this product in preruminating calves. Do not use in calves to be processed for veal.
Human Warnings
For use in animals only. Keep out of reach of children. Avoid contact with eyes. In case of contact, immediately flush eyes with copious amounts of water for 15 minutes. In case of dermal contact, wash skin with soap and water. Consult a physician if irritation persists following ocular or dermal exposures. Individuals with a history of hypersensitivity to quinolones should avoid this product. In humans, there is a risk of user photosensitization within a few hours after excessive exposure to quinolones. If excessive accidental exposure occurs, avoid direct sunlight. To report adverse reactions or to obtain a copy of the Safety Data Sheet (SDS), call 1-888-963-8471.
Precautions
The effects of danofloxacin on bovine reproductive performance, pregnancy, and lactation have not been determined.
Subcutaneous injection can cause a transient local tissue reaction that may result in trim loss of edible tissue at slaughter.
Quinolone-class drugs should be used with caution in animals with known or suspected central nervous system (CNS) disorders. In such animals, quinolones have, in rare instances, been associated with CNS stimulation, which may lead to convulsive seizures.
Quinolone-class drugs have been shown to produce erosions of cartilage of weight-bearing joints and other signs of arthropathy in immature, rapidly growing animals of various species. Refer to Animal Safety for information specific to danofloxacin.
Adverse Reactions
A hypersensitivity reaction was noted in 2 healthy calves treated with ADVOCIN in a laboratory study. In one location of a multi-site field trial, one out of the 41 calves treated with 6 mg/kg q 48 hours showed lameness on Day 6 only. In this same field trial location one of 38 calves treated with 8 mg/kg once became lame 4 days after treatment and remained lame on the last day of the study (Day 10). Another calf in the same treatment group developed lameness on the last day of the study.
(A) Pharmacokinetics
Danofloxacin distributes extensively throughout the body, as evidenced by a steady state volume of distribution (VDss) in cattle exceeding 1 L/kg. Danofloxacin concentrations in the lung homogenates markedly exceed those observed in plasma, further suggesting extensive distribution to the indicated site of infection. Danofloxacin is rapidly eliminated from the body (apparent terminal elimination T. ranging from 3–6 hours), and negligible accumulation was observed when animals were dosed twice, 48 hours apart.
Danofloxacin is rapidly absorbed and is highly bioavailable when administered as a subcutaneous injection in the neck. Linear pharmacokinetics has been demonstrated when danofloxacin is administered to cattle by subcutaneous injection at doses between 1.25 to 10 mg/kg.
No statistically significant gender difference was observed in peak or total systemic exposure following a single subcutaneous administration of danofloxacin to heifers and steers at a dose of 6 mg/kg body weight (Table 1).
| Steers | Heifers | ||||
|---|---|---|---|---|---|
| Mean | %CV Coefficient of variation % | Mean | %CV | ||
| aAUC0-24 | µg × hr/mL | 9.4 | 10 | 8.8 | 9 |
| bF% | 92 | 5 | 87 | 3 | |
| aCmax | µg/mL | 1.25 | 16 | 1.27 | 13 |
| a,cTmax | hr | 3.2 | 42 | 1.7 | 31 |
| dCL | L/hr | 0.54 | 12 | 0.62 | 9 |
| dVDss | L/kg | 2.7 | 7 | 2.6 | 4 |
| aT½ | hr | 4.8 | 18 | 4.2 | 7 |
a AUC0-24 = area under the plasma concentration versus time curve from hr zero to hr 24 postdose. Cmax = maximum observed concentration. Tmax = time to Cmax.
b Bioavailability (F%) = extent of drug absorption following subcutaneous administration. Within subject F values were determined as the ratio of AUC0-inf values estimated following a 6 mg/kg dose administered by either a subcutaneous or intravenous injection.
c Statistically significant differences in Tmax were detected between genders. Given the similarity in Cmax values, these differences are not expected to have any clinical relevance.
d Clearance (CL) and Volume of distribution at steady state (VDss) were determined from data obtained after intravenous administration of a 6 mg/kg dose.
e Coefficient of variation %
(B) Microbiology
Danofloxacin exerts its activity by inhibiting the bacterial DNA gyrase enzyme, thereby blocking DNA replication. Inhibition of DNA gyrase is lethal to bacteria and danofloxacin has been shown to be rapidly bactericidal. Danofloxacin is active against gram-negative and gram-positive bacteria.
The Minimum Inhibitory Concentrations (MIC) of danofloxacin for pathogens isolated in natural infections from various clinical studies in North America, 1996–1997, were determined using the standardized microdilution technique (Sensititre/Alamar, Accumed International), and are shown in Table 2
Table 2. Danofloxacin minimum inhibitory concentration (MIC) values* of indicated pathogens isolated from 1996-1997 pivotal BRD treatment field studies in the U.S. | ||||
Indicated Pathogen | Number of Isolates | MIC50** (μg/ mL) | MIC90** (μg/ mL) | MIC Range (μg/ mL) |
Mannheimia haemolytica | 106 | 0.06 | 0.06 | ≤0.015 to 0.12 |
Pasteurella multocida | 94 | ≤0.015 | 0.06 | ≤0.015 to 0.12 |
* The correlation between in vitro susceptibility data and clinical effectiveness is unknown. Table 3. Danofloxacin minimum inhibitory concentration (MIC) values* of indicated pathogens isolated from 2013 pivotal field studies in the U.S. and Canada for the control of BRD in cattle at high risk of developing BRD. | ||||
Indicated Pathogen | Number of Isolates | MIC50** (μg/ mL) | MIC90** (μg/ mL) | MIC Range (μg/ mL) |
Mannheimia haemolytica | 507 | 0.03 | 0.06 | ≤0.008 to >8 |
Pasteurella multocida | 324 | ≤0.008 | 0.12 | ≤0.008 to 1.0 |
*The correlation between in vitro susceptibility data and clinical effectiveness is unknown.
**The lowest MIC to encompass 50% and 90% of the most susceptible isolates, respectively.
Effectiveness
The effectiveness of 8 mg/kg administered once and the 6 mg/kg BW alternate day regimen was confirmed in 4 well-controlled studies of naturally acquired bacterial respiratory infections in feedlot age cattle. These studies were conducted under commercial conditions at 4 locations in North America. Bacterial pathogens isolated in the clinical field trial are provided in the Microbiology section.
The effectiveness of ADVOCIN for the control of BRD in cattle at high risk of developing BRD associated with Mannheimia haemolytica and Pasteurella multocida was demonstrated in a multi-site study conducted in North America. The study enrolled a total of 1,480 commercial, crossbred-beef, Holstein and Holstein-cross steer calves at high risk of developing BRD associated with M. haemolytica and P. multocida.
At enrollment, calves were randomly administered a one-time subcutaneous injection of either ADVOCIN at a dosage rate of 8 mg/kg of body weight or an equivalent volume of sterile saline. Cattle were observed daily for clinical signs of BRD and were evaluated for clinical success on Day 10 post-treatment. The treatment success rate of ADVOCIN-treated calves (86.0%) was statistically significantly (p=0.0068) greater than that of saline-treated calves (76.3%) (based on back-transformed least squares means). No adverse events associated with ADVOCIN administration were reported in the study.
Animal Safety
Safety studies were conducted in feeder calves using single doses of 10, 20, or 30 mg/kg for 6 consecutive days and 18, 24, or 60 mg/kg for 3 consecutive days. No clinical signs of toxicity were observed at doses of 10 and 20 mg/kg when administered for 6 days, nor at doses of 18 and 24 mg/kg when administered for 3 days. Articular cartilage lesions, consistent with fluoroquinolone chondropathy, were observed after examination of joints from animals as follows: one of 5 animals administered 18 mg/kg for 3 days; one of 6 animals administered 20 mg/kg or 6 days; 5 of 6 animals administered 0 mg/kg for 6 days; and in all 4 animals administered 60 mg/kg for 3 days. Clinical signs of inappetence, transient lameness (2/6), ataxia (2/6), tremors (2/6), nystagmus (1/6), exophthalmos (1/6), and recumbency (2/6) were observed when a dose of 30 mg/kg was administered for 6 consecutive days. Recumbency and depression were seen in one out of 4 animals administered 60 mg/kg for 3 days. Swelling at the injection site was noted at each dose level.
Safety was also evaluated in 21-day-old calves. In one group, these immature animals were given injections of 6 mg/kg on study days 0, 2, 3, 5, 6, and 8. A second group of animals received injections of 18 mg/kg for a total of 2 injections 48 hours apart. The only treatment-related sign was erythema of the nasal pad in 3 of 6 calves that received 18 mg/kg. One calf in the 6 mg/kg group had pretreatment scleral erythema, and developed nasal erythema after treatment that may or may not have been treatment-related. No changes in clinical pathology parameters were observed. No articular cartilage lesions were observed in the joints at any dosage.
An injection site study conducted in feeder calves demonstrated that the product can induce a transient local reaction in the subcutaneous tissue and underlying tissue.
Toxicology
Ninety-day oral toxicity studies in dogs and rats established a no observable effect level (NOEL) of 2.5 mg/ kg bw/day and 2.4 mg/kg bw/day, respectively. Higher doses in juvenile dogs produced arthropathy, a typical quinolone-associated side effect. In chronic rodent bioassays, no evidence of carcinogenicity was associated with long-term danofloxacin administration in rats and mice. No teratogenic effects were observed in rodents at doses up to 50 mg/kg bw/day (mice) or 100 mg/kg bw/ day (rats) or in rabbits at the highest dose tested of 15 mg/kg bw/day. A three-generation rat reproductive toxicity study established a NOEL of 6.25 mg/kg bw/day. Microbial safety analyses indicate that danofloxacin residues present in edible tissues of treated animals under the current use conditions would most likely not cause adverse effects on the human intestinal microflora of the consumer.
Storage Information
Store at or below 30°C (86°F). Use this product within 28 days of the first puncture and puncture a maximum of 7 times. If more than 7 punctures are anticipated, the use of automatic injection equipment or a repeater syringe is recommended. When using a draw-off spike or needle with bore diameter larger than 16 gauge, discard any product remaining in the vial immediately after use. Protect from light. Protect from freezing. The color is yellow to amber and does not affect potency.
How Supplied
ADVOCIN (180 mg danofloxacin/mL) is supplied in 100- and 250-mL, amber-glass, sterile, multi-dose vials.
Approved by FDA under NADA # 141-207
zoetis
Distributed by:
Zoetis Inc.Kalamazoo, MI 49007
Use Only as Directed
Contact Information
To report suspected adverse effects and/or obtain a copy of the SDS or for technical assistance, call Zoetis Inc. at
1-888-963-8471.
For additional information about adverse drug experience reporting for animal drugs, contact FDA at 1-888-FDA-VETS
or online at www.fda.gov/reportanimalae.
| TAKE TIME | OBSERVE LABEL DIRECTIONS |
Revised: October 2020
40032874
40032877
Principal Display Panel - 100 Ml Bottle Label
Principal Display Panel - 250 Ml Bottle Label
* The information on this page is for an ANIMAL PRODUCT, please review the complete disclaimer below.