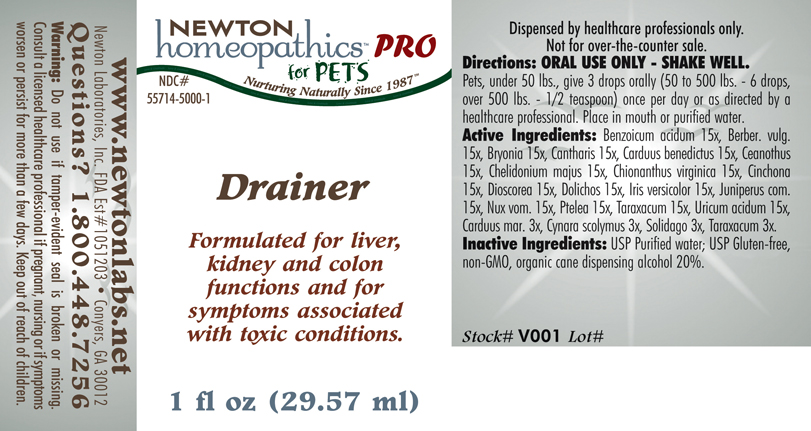
Animal NDC 55714-5048-1 Drainer
Benzoicum Acidum, Berber. Vulg., Bryonia, Cantharis, Carduus Benedictus, Ceanothus, Chelidonium Majus, Chionanthus Virginica, Cinchona, Dioscorea, Dolichos, Iris Versicolor, Juniperus Com., Nux Vom., Ptelea, Taraxacum, Uricum Acidum, Carduus Mar., Cynara Scolymus, Solidago
Animal Product Information
| Field Name | Field Value |
|---|---|
| Animal NDC Code | 55714-5048-1 |
| Proprietary Name | Drainer What is the Proprietary Name? The proprietary name also known as the trade name is the name of the product chosen by the medication labeler for marketing purposes. |
| Non-Proprietary Name | Benzoicum Acidum, Berber. Vulg., Bryonia, Cantharis, Carduus Benedictus, Ceanothus, Chelidonium Majus, Chionanthus Virginica, Cinchona, Dioscorea, Dolichos, Iris Versicolor, Juniperus Com., Nux Vom., Ptelea, Taraxacum, Uricum Acidum, Carduus Mar., Cynara Scolymus, Solidago What is the Non-Proprietary Name? The non-proprietary name is sometimes called the generic name. The generic name usually includes the active ingredient(s) of the product. |
| Labeler Name | Newton Laboratories, Inc. |
| Product Type | Otc Animal Drug |
| Usage Information |
|
| Active Ingredient(s) |
|
| Inactive Ingredient(s) |
|
| Marketing Category | UNAPPROVED HOMEOPATHIC What is the Marketing Category? Product types are broken down into several potential Marketing Categories, such as NDA/ANDA/BLA, OTC Monograph, or Unapproved Drug. One and only one Marketing Category may be chosen for a product, not all marketing categories are available to all product types. Currently, only final marketed product categories are included. The complete list of codes and translations can be found at www.fda.gov/edrls under Structured Product Labeling Resources. |
Drainer Images
Drainer Animal Product Labeling Information
The product labeling information includes all published material associated to a drug. Product labeling documents include information like generic names, active ingredients, ingredient strength dosage, routes of administration, appearance, usage, warnings, inactive ingredients, etc.
Table of Contents
Indications & Usage Section
Formulated for symptoms associated with toxicity such as fatigue and sluggish elimination.
Dosage & Administration Section
Directions: Pets, under 50 lbs., give 3 drops orally (50 to 500 lbs. - 6 drops, over 500 lbs. - 1/2 teaspoon) once per day or as directed by a health professional. Place in mouth or purified water.
Active Ingredient Section
Equal parts of Cynara 6x, Solidago 6x, Taraxacum 6x, Benzoicum ac. 15x, Berber. vulg. 15x, Bryonia 15x, Cantharis 15x, Carduus ben. 15x, Carduus mar. 15x, Ceanothus 15x, Chelidonium maj. 15x, Chionanthus 15x, Cinchona 15x, Dioscorea 15x, Dolichos 15x, Iris vers. 15x, Juniperus com.15x, Nux vom. 15x, Ptelea 15x, Taraxacum 15x, Uricum ac. 15x.
Inactive Ingredient Section
Inactive Ingredients: USP Purified Water; USP Gluten-free, non-GMO, organic cane alcohol 20%.
Questions Section
newtonlabs.net Questions? 800.448.7256
Newton Laboratories, Inc. FDA Est # 1051203 - Conyers, GA 30013
Warnings Section
Warnings:Keep out of reach of children. Do not use if tamper - evident seal is missing. If symptoms worsen or persist for more than a few days, consult a doctor. If pregnant or breast-feeding, ask a doctor before use.
Pregnancy Or Breast Feeding Section
If pregnant or breast-feeding, ask a doctor before use.
Keep Out Of Reach Of Children Section
Keep out of reach of children.
Package Label
* The information on this page is for an ANIMAL PRODUCT, please review the complete disclaimer below.