Animal NDC 58198-0369-2 Component Te-g With Tylan
Trenbolone Acetate, Estradiol, Tylosin Tartrate
Animal Product Information
Component Te-g With Tylan Animal Product Labeling Information
The product labeling information includes all published material associated to a drug. Product labeling documents include information like generic names, active ingredients, ingredient strength dosage, routes of administration, appearance, usage, warnings, inactive ingredients, etc.
Table of Contents
Other
Elanco™
Component™
TE-G with Tylan™
(trenbolone acetate and estradiol and tylosin tartrate implants)
FOR GROWING BEEF STEERS AND HEIFERS ON PASTURE (Stocker, Feeder, and Slaughter)
Caution: Federal law restricts this drug to use by or on the order of a licensed veterinarian.
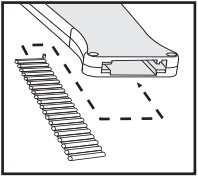
20 dose Cartridge Belt
Use with a Component Implanter
Description:
Each cartridge belt holds 20 doses of Component TE-G with Tylan (trenbolone acetate and estradiol and tylosin tartrate implant) Implants. Each dose of 3 pellets consists of 2 pellets containing a total of 40 mg of trenbolone acetate and 8 mg estradiol plus 1 pellet containing 29 mg tylosin tartrate as a local antibacterial.
Indications For Use:
For increased rate of weight gain in growing beef steers and heifers on pasture (stocker, feeder, and slaughter).
Not approved for repeated implantation (re-implantation) with this or any other cattle ear implant in growing beef steers and heifers on pasture (stocker, feeder, and slaughter). Safety and effectiveness following re-implantation have not been evaluated.
Do not use in beef calves less than 2 months of age, dairy calves, and veal calves because effectiveness and safety have not been established.
Do not use in animals intended for subsequent breeding, or in dairy cows.
Warning:
This product is intended for use in cattle at high risk of developing ear abscesses. Not recommended for use in cattle at low risk of developing ear abscesses (e.g., dry, clean cattle).
The benefit of using a cattle ear implant containing a tylosin tartrate pellet when animals receive a concomitant antimicrobial product has not been evaluated.
Withdrawal Periods and Residue Warnings
No withdrawal period is required when used according to labeling.
Do not use in beef calves less than 2 months of age, dairy calves, and veal calves. A withdrawal period has not been established for this product in pre-ruminating calves.
Do not use in dairy cows or in animals intended for subsequent breeding. Use in these cattle may cause drug residues in milk and/or calves born to these cows.
Implant pellets subcutaneously in ear only. Any other location is a violation of Federal law. Do not attempt salvage of implanted site for human or animal food.
User Safety Warnings:
Not for human use. Keep out of reach of children.
Restricted Drug (California) - use only as directed.
Implanting Instructions:
Loading the Implanter
Load the implanter following the instructions supplied with each implanter.
Restrain the Animal
Speed of implantation as well as safety of handlers is best achieved by restraining animal in a squeeze chute using head restraint. When implanting horned cattle, better control is obtained with additional use of nose tongs.
Prepare the Implant Site
Scrub the back side of the ear (implant site) with a piece of clean absorbent cotton which has been soaked with topical germicidal solution. Follow manufacturer’s directions on germicide for correct strength and preparation of solution. Avoid getting disinfectant in animal’s eyes.
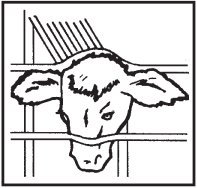
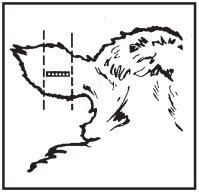
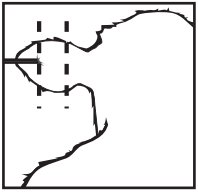
Where to Implant
The full contents of one cartridge cell should be implanted beneath the skin on the back side of the middle one-third of the ear as illustrated in the drawing. The implant must not be closer to the head than the edge of the auricular cartilage ring farthest from the head. The location for insertion of the needle is a point toward the tip of the ear at least a needle length away from the intended deposition site. Avoid injuring the large arteries, veins and cartilage of the ear.
Insert the Needle
With one hand firmly grasp the ear. With the other hand insert needle point through the skin and ease forward on a lateral plane until the entire length of the needle is under the skin.
Implant the Pellets
After inserting the needle fully in the correct position, squeeze the trigger fully as the needle is withdrawn from the ear. This properly deposits the implant in the needle track. This procedure should prevent breakage or crushing of pellets if otherwise forced into contact with tough fibrous-tissue underlying the skin. The length and total contact area of the single dose are designed to permit absorption of the hormones after implantation to stimulate good weight gain. Broken or crushed pellets may interfere with rates of gain.
Storage Conditions
Store unopened product at or below 25°C (77°F). Avoid excessive heat and humidity.
Use before the expiration date printed on foil pouch. Discard open foil pouches.
Approved by FDA under ANADA # 200-221
Manufactured by a non-sterilizing process.
Component TE-G with Tylan is covered by U.S. Patent No. 5,874,098
Component, Tylan, Elanco and the diagonal bar logo are trademarks of Elanco or its affiliates.
© 2023 Elanco or its affiliates
Distributed by Elanco US Inc,
Greenfield, IN 46140, USA
Tylosin: Product of the United Kingdom
QUESTIONS/COMMENTS? For a copy of the Safety Data Sheet or to report side effects, contact Elanco US, Inc. at 1-888-545-5973. For additional information about adverse drug experience reporting for animal drugs, contact FDA at 1-888-FDA-VETS or http://www.fda.gov/reportanimalae.
April 2023
PA104024X AH0369
Principal Display Panel - Cartridge Label
Elanco™
Component™
TE-G with Tylan™
™
(trenbolone acetate and estradiol and tylosin tartrate implants)
For Growing Beef Steers and Heifers on
Pasture (Stocker, Feeder, and Slaughter)
One 20-Dose
Cartridge Belt
Principal Display Panel - Carton Label
Elanco™
Component™
TE-G with Tylan™
™
(trenbolone acetate and estradiol and tylosin tartrate implants)
For Growing Beef Steers and Heifers on
Pasture (Stocker, Feeder, and Slaughter)
* The information on this page is for an ANIMAL PRODUCT, please review the complete disclaimer below.