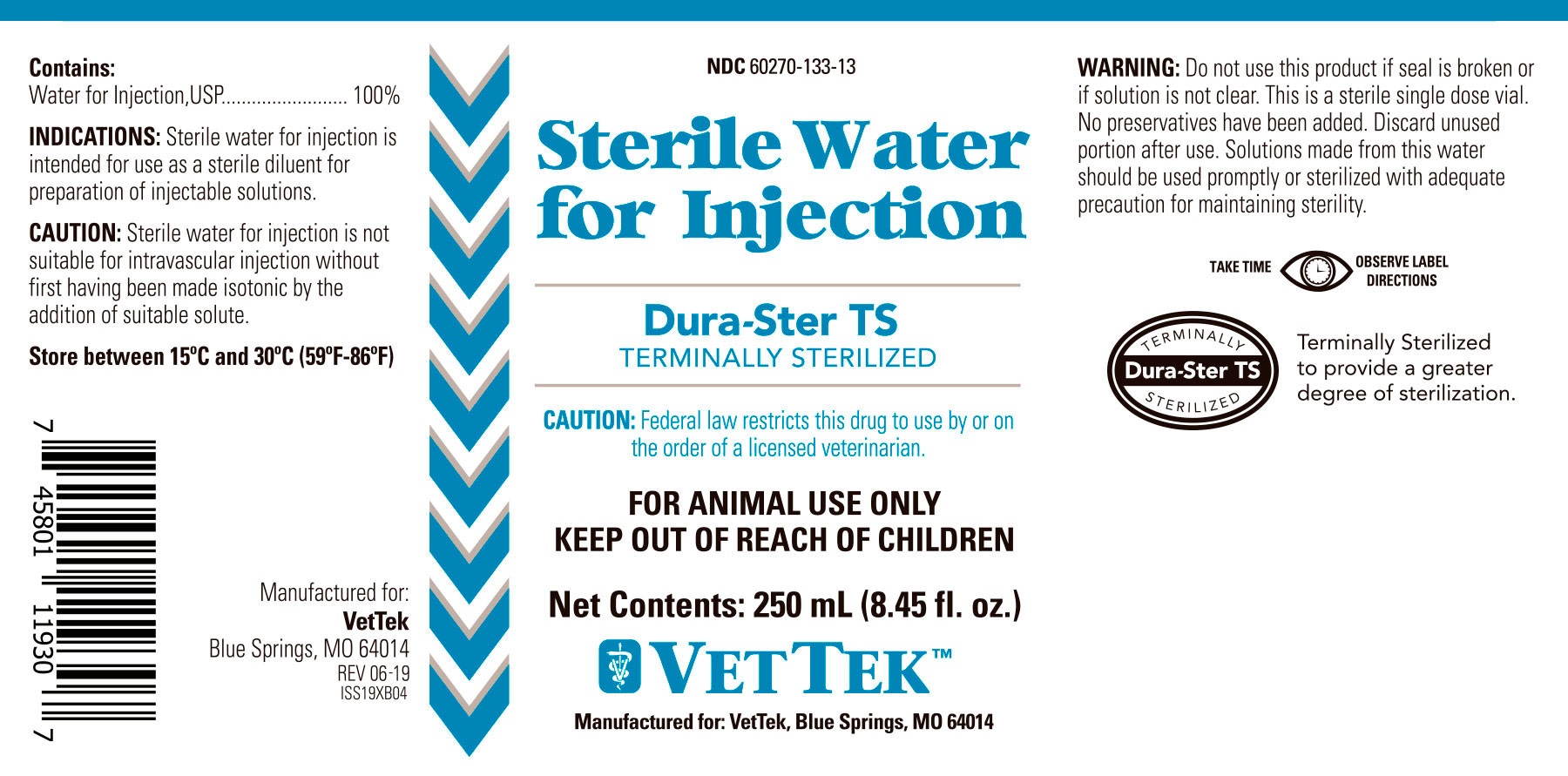
Animal NDC 60270-133-13 Sterile Water
Sterile Water For Injection
Animal Product Information
Sterile Water Images
Sterile Water Animal Product Labeling Information
The product labeling information includes all published material associated to a drug. Product labeling documents include information like generic names, active ingredients, ingredient strength dosage, routes of administration, appearance, usage, warnings, inactive ingredients, etc.
Table of Contents
Other
STERILE WATER- sterile water for injection
Active Ingredient
Contains:
Water For Injection, USP................ 100%
Indications:
Sterile water for injection is intended for use as a sterile diluent for preparation of injectable solutions.
Caution:
Sterile water for injection is not suitable for intravascular injection without first having been made isotonic by the addition of suitable solute.
Federal law restricts this drug to use by or on the order of a licensed veterinarian.
Storage
Store between 15°C and 30°C (59°F-86°F).
Veterinary Indications
KEEP OUT OF REACH OF CHILDREN
Warning:
Do not use this product if seal is broken or if solution is not clear. This is a sterile single dose vial. No preservatives have been added. Discard unused portion after use. Solutions made from this water should be used promptly or sterilized with adequate precautions for maintaining sterility.
Dosage & Administration
TAKE TIME OBSERVE LABEL DIRECTIONS.
Terminally Sterilized to provide a greated degree of sterilization.
Information For Owners/Caregivers
Manufactured for:
VetTek
Blue Springs, MO, 64014
REV 06-19
ISS19XB04
Principal Display Panel
NDC 60270-133-13
Sterile Water for Injection
Dura-Ster TS
TERMINALLY STERILIZED
CAUTION: Federal law restricts this drug to use by or on the order of a licensed veterinarian.
FOR ANIMAL USE ONLY
KEEP OUT OF REACH OF CHILDREN
Net Contents: 250 mL (8.45 fl. oz.)
VETTEKTM
Manufactured for: VetTek, Blue Springs, MO, 64014
* The information on this page is for an ANIMAL PRODUCT, please review the complete disclaimer below.