Pill Identifier Prevacid - NDC 64764-541
Product Code 64764-541
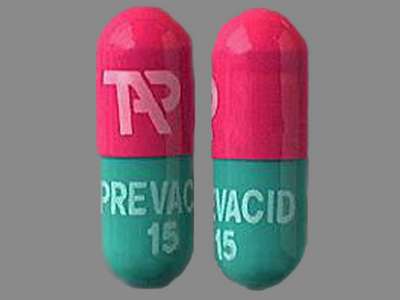
Prevacid by Takeda Pharmaceuticals America, Inc. is a green and pink capsule about 16 mm in size, imprinted with tap;prevacid;15. The product is a with active ingredient(s) .
Prevacid
Active Ingredient(s):
Inactive Ingredient(s):
- sucrose
- methacrylic acid - ethyl acrylate copolymer (1:1) type A
- low-substituted hydroxypropyl cellulose, unspecified
- starch, corn
- magnesium carbonate
- talc
- polyethylene glycol, unspecified
- titanium dioxide
- polysorbate 80
- hydroxypropyl cellulose (1600000 wamw)
- silicon dioxide
- D&C Red No. 28
- FD&C Blue No. 1
- FD&C Red No. 40
- FD&C Green No. 3
Takeda Pharmaceuticals America, Inc.
Product Type:
Dosage Form:
- Green (C48329)
- Pink (C48328)
Product Packages
| NDC Package Code | Package Description |
|---|---|
| 64764-541-05 | 5 BLISTER PACK in 1 TRAY / 4 CAPSULE, DELAYED RELEASE in 1 BLISTER PACK |
| 64764-541-11 | 10 BLISTER PACK in 1 CARTON / 10 CAPSULE, DELAYED RELEASE in 1 BLISTER PACK |
| 64764-541-19 | 1000 CAPSULE, DELAYED RELEASE in 1 BOTTLE |
| 64764-541-30 | 30 CAPSULE, DELAYED RELEASE in 1 BOTTLE |
* Please review the disclaimer below.
Pill Identification data last updated on: December, 2018
This product uses publicly available data from the U.S. National Library of Medicine (NLM), National Institutes of Health, Department of Health and Human Services; NLM is not responsible for the product and does not endorse or recommend this or any other product. If this is an emergency, call 911 or the national Poison Help hotline at 1-800-222-1222. Most images are not part of the product labels and have not been verified by the labeler company. ALWAYS consult your physician or pharmacist with any medication-related questions.