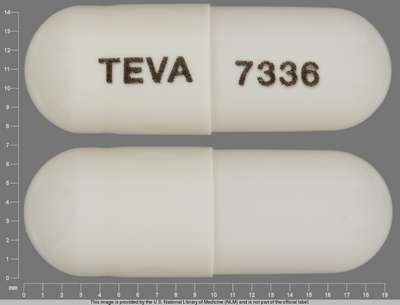
Pill Identifier Topiramate - NDC 0093-7336
Topiramate
Product Code 0093-7336
Topiramate by Teva Pharmaceuticals Usa, Inc. is a white capsule capsule coated pellets about 19 mm in size, imprinted with teva;7336. The product is a human prescription drug with active ingredient(s) topiramate.
Topiramate
Active Ingredient(s): Topiramate
Inactive Ingredient(s):
- FERROSOFERRIC OXIDE
- ETHYLCELLULOSE (7 MPA.S)
- GELATIN, UNSPECIFIED
- POVIDONE K30
- POLYETHYLENE GLYCOL 6000
- PROPYLENE GLYCOL
- SHELLAC
- SUCROSE
- STARCH, CORN
- TALC
- TITANIUM DIOXIDE
- TRIETHYL CITRATE
- POTASSIUM HYDROXIDE
- AMMONIA
Teva Pharmaceuticals Usa, Inc.
Product Type: Human Prescription Drug
Dosage Form: Capsule, Coated Pellets
- White (C48325)
Product Packages
| NDC Package Code | Package Description |
|---|---|
| 0093-7336-06 | 60 CAPSULE, COATED PELLETS in 1 BOTTLE |
* Please review the disclaimer below.
Pill Identification data last updated on: December, 2018
This product uses publicly available data from the U.S. National Library of Medicine (NLM), National Institutes of Health, Department of Health and Human Services; NLM is not responsible for the product and does not endorse or recommend this or any other product. If this is an emergency, call 911 or the national Poison Help hotline at 1-800-222-1222. Most images are not part of the product labels and have not been verified by the labeler company. ALWAYS consult your physician or pharmacist with any medication-related questions.