Product Images Trulicity
View Photos of Packaging, Labels & Appearance
- Logo - logo
- QR Code - qr v1
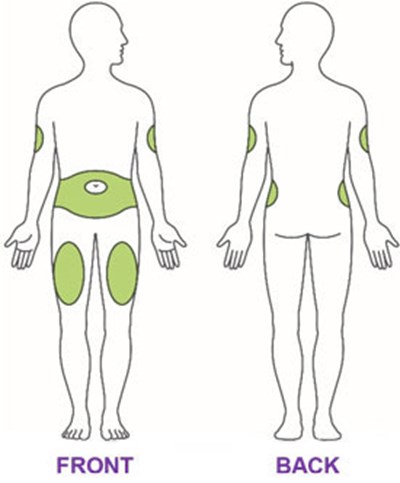
- Figure - trulicity ifu 0point75 injection sites v1
- Figure - trulicity ifu 1point5 injection sites v1
- Figure - trulicity ifu 3mg injection sites v1
- Figure - trulicity ifu 4point5mg injection sites v1
- Figure - trulicity ifu check v1
- Exclamation - trulicity ifu exclamation v1
- Figure - trulicity ifu expiration location v2
- Figure - trulicity ifu inspect v1
- Locked - trulicity ifu locked v1
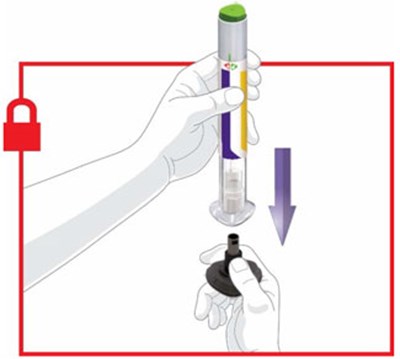
- Figure - trulicity ifu prepare v1
- Figure - trulicity ifu remove v1
- Figure - trulicity ifu sup 0point75 f2 v3
- Figure - trulicity ifu sup 0point75 f3 v3
- Figure - trulicity ifu sup 0point75 f4 v3
- Figure - trulicity ifu sup 0point75 f5 v3
- Figure - trulicity ifu sup 0point75 pen parts v3
- Figure - trulicity ifu sup 0point75 pen v3
- Figure - trulicity ifu sup 1point5 f2 v3
- Figure - trulicity ifu sup 1point5 f3 v3
- Figure - trulicity ifu sup 1point5 f4 v3
- Figure - trulicity ifu sup 1point5 f5 v3
- Figure - trulicity ifu sup 1point5 pen parts v3
- Figure - trulicity ifu sup 1point5 pen v3
- Figure - trulicity ifu sup 3mg f2 v1
- Figure - trulicity ifu sup 3mg f3 v1
- Figure - trulicity ifu sup 3mg f4 v1
- Figure - trulicity ifu sup 3mg f5 v1
- Figure - trulicity ifu sup 3mg pen parts v1
- Figure - trulicity ifu sup 3mg pen v1
- Figure - trulicity ifu sup 4point5 f2 v1
- Figure - trulicity ifu sup 4point5 f3 v1
- Figure - trulicity ifu sup 4point5 f4 v1
- Figure - trulicity ifu sup 4point5 f5 v1
- Figure - trulicity ifu sup 4point5 pen parts v1
- Figure - trulicity ifu sup 4point5 pen v1
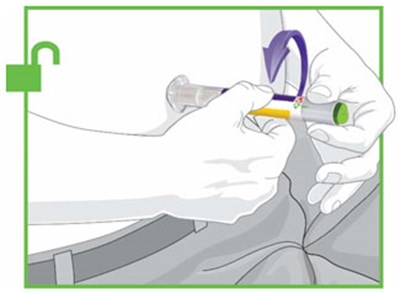
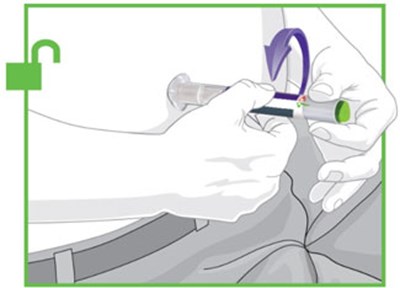
- Figure - trulicity ifu unfold v1
- Unlocked - trulicity ifu unlock v1
- Figure 1 - trulicity pi f1 v2
- Figure 2 - trulicity pi f2 v2
- Figure 3 - trulicity pi f3 v1
- Figure 5 - trulicity pi f4 v1
- Figure 6 - trulicity pi f5 v1
- Figure 4 - trulicity pi f6 v1
- us trulicity sup 1point5mg 005 04
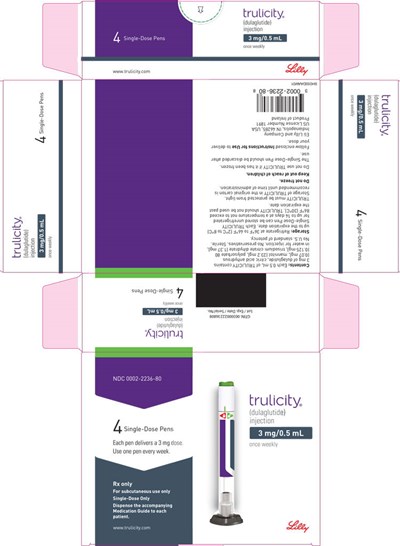
- PACKAGE LABEL – Trulicity™, 3 mg/0.5 mL, Prefilled Pen, 4 count - us trulicity sup 3mg 055 01
- PACKAGE LABEL – Trulicity™, 4.5 mg/0.5 mL, Prefilled Pen, 4 count - us trulicity sup 4point5mg 056 01
- us trulicity sup point75mg 004 04
Product Label Images
The following 49 images provide visual information about the product associated with Trulicity NDC 0002-2236 by Eli Lilly And Company, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
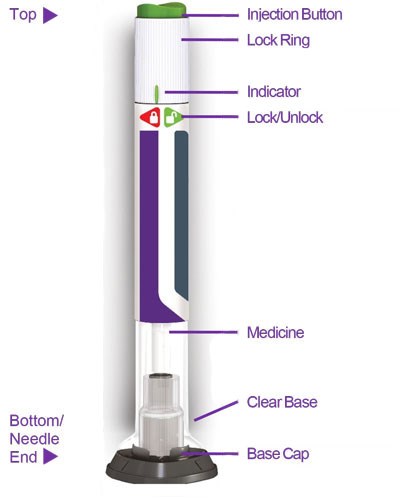
Figure - trulicity ifu sup 0point75 pen parts v3

This is a diagram of a medical injection device. It includes a "Top" section with an injection button, lock ring, and an indicator. There is also a "Bottom" section with a clear base, needle end, and base cap. The text also includes a "Lock/Unlock" feature accessed with "QF=".*
Figure - trulicity ifu sup 1point5 pen parts v3

The given text is a description of a medical injection device. The device comprises a clear base at the bottom, a lock ring, an injection button at the top "b mam," and an indicator. The device has a provision for locking and unlocking, and also facilitates medication through the injection of a needle. No further information can be extracted from the given text.*
Figure - trulicity ifu sup 3mg f4 v1

This text describes the process of administering an injection and indicates that the completion of the injection is indicated by the visibility of the gray plunger.*
Figure - trulicity ifu sup 4point5 f4 v1

The text is a set of instructions to indicate how to know when an injection is complete. It says that the injection is finished when the gray plunger is visible. However, there is not enough information to determine what type of injection or medication is being referred to or any other details.*
Figure - trulicity ifu sup 4point5 pen parts v1
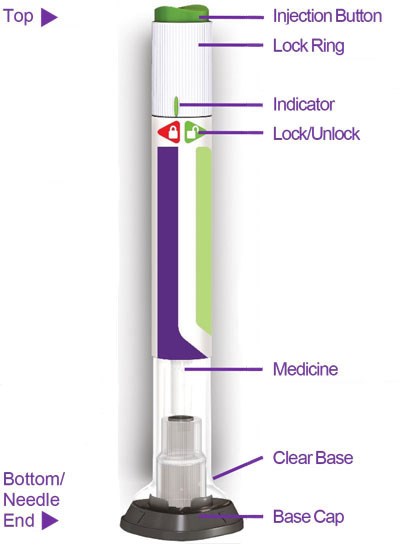
This appears to be a description of a medical device, possibly an injection device. It includes labels for various parts of the device, including a "Top p MR Injection Button" and a "Needle 4," as well as mention of a "Medicine" section and a "ClearBase." There are also references to a "LockRing," "Indicator," and "LockUniock." Without further context it is difficult to provide a more specific description or use for this device.*
Figure 4 - trulicity pi f6 v1

The text describes different dosages of Trulicity administered to a group of subjects over the course of several weeks. It lists the number of subjects and their observed data at different intervals and provides information about the Observed mean HbATc levels at scheduled visits and retrieved dropout multiple imputation (MI) based estimates at week 36.*
us trulicity sup 1point5mg 005 04

This text appears to be partial information related to a medication called Trulicity which comes in the form of an injection with 1.5 mg/0.5 mL dose. The text also suggests using one pen every week, however, it is unclear why or for what condition this medication is used.*
PACKAGE LABEL – Trulicity™, 4.5 mg/0.5 mL, Prefilled Pen, 4 count - us trulicity sup 4point5mg 056 01
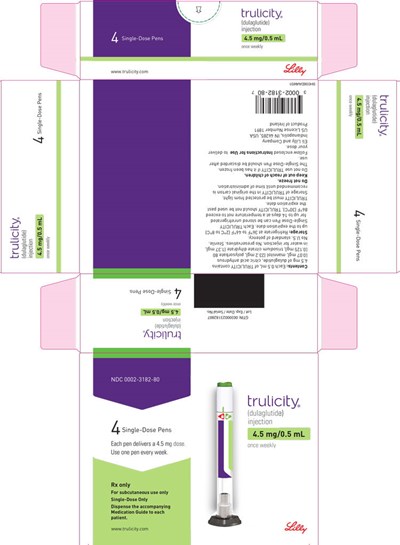
This is a description of a medication called Trulicity (dulaglutide), which is administered through a single-dose pen delivering a 4.5mg/0.5mL dose to be used subcutaneously once a week.*
us trulicity sup point75mg 004 04

Trulicity is a medication delivered via a single-dose pen containing 0.75mg/0.5 mL of Dulaglutide that is recommended for subcutaneous use once every week. The medication is prescribed for the treatment of type 2 diabetes.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.