Product Images Cyramza
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 10 images provide visual information about the product associated with Cyramza NDC 0002-7669 by Eli Lilly And Company, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
PACKAGE CARTON – CYRAMZA 100 mg/10 mL single-use vial - cyramza 100mg carton 001 02

Cyramza (ramucirumab) is an injection used for cancer treatment. The storage conditions require refrigeration between 2°C-5°C and must not be frozen or shaken. It comes in single-dose vials and should be discarded if not used. The concentration of the drug is 100mg/10mL and should not be used after the expiration date.*
PACKAGE CARTON – CYRAMZA 100 mg/50 mL single-use vial - cyramza 100mg carton 003 01

Cyramza is a medication injected with 100mg/10mL concentration (110 mg/mL). It is recommended to store in refrigeration between 2-6 °C (36-46°F) and avoid exposure to direct light. The dosage and administration depend on the patient's condition and must be followed according to the instructions in the package. Cyramza contains 100 mg of ramucirumab, 998 mg of L-histidine monohydrochloride (.65% w/v), polysorbate 80 0.1 mg/mL, sodium chloride 420 mg/mL. The product is produced by Lilly and Company in Indianapolis. No other information is available.*
PACKAGE CARTON – CYRAMZA 500 mg/50 mL single-use vial - cyramza 500mg carton 000 02

Cyramza® Injection is a medication that comes in a 500mg/50mL dosage and contains 10mg/mL Famucirumab. It should be stored at 2°C to 5°C and should not be frozen or shaken. The medication should be prescribed only by authorized medical professionals and should be administered only to women suffering from certain conditions. This text also contains lot number information, but it is not easily readable.*
PACKAGE CARTON – CYRAMZA 500 mg/50 mL single-use vial - cyramza 500mg carton 004 01

This is a medication package insert for an injection called Cyramza®. The product contains Famucirumab and comes in a 500mg/50mL solution for injection. The dosage instructions are included as well as storage recommendations. The text also contains information about ingredients, precautions, and contact information for the manufacturer.*
Figure 1 - cyramza pi f001 v2
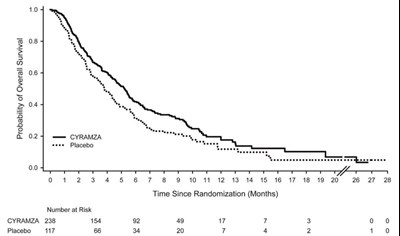
This appears to be a graph depicting the probability of overt events over time since randomization (in months). The graph includes two lines, one for treatment with CYRAMZA and one for treatment with a placebo. The x-axis appears to go up to 28 months and the y-axis shows the probability percentage from 0 to 10%.*
Figure 2 - cyramza pi f002 v2
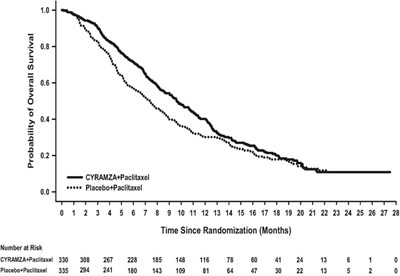
This appears to be a medical document displaying test results for drug CYRAMZA when administered with Paclitaxel. The table represents the number of participants at risk for a certain time period since beginning the treatment.*
Figure 4 - cyramza pi f004 v2
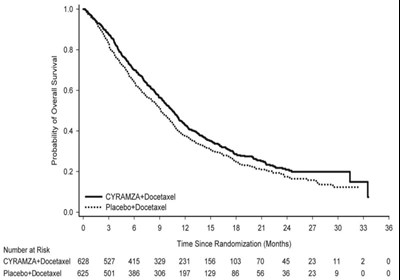
This is a graph showing the probability of overall survival for two groups of patients undergoing treatment with either CYRAMZA+Docetaxel or Placebo+Docetaxel. The "Number at Risk" table indicates the number of patients still alive and participating in the study at specific time intervals since randomization. The graph shows that patients treated with CYRAMZA+Docetaxel had a higher probability of overall survival than those treated with Placebo+Docetaxel.*
Figure 5 - cyramza pi f005 v2
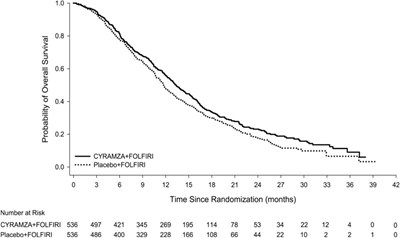
This appears to be a graph showing the Probability of Overall Survival over Time Since Randomization (months) for two different treatments, CYRAMZA+FOLFIRI and Placebo+FOLFIRI. The text on the left shows the Number at Risk for each treatment at different time intervals.*
Figure 6 - cyramza pi f006 v2

This is a survival curve depicting the probability of overall survival for two treatments, CYRAMZA + BSC and Placebo + BSC, measured over a period of 24 months. The graph also provides information on the number of participants at risk and the time since randomization. The exact context and purpose of the study that generated the data is not available.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.
