Product Images Opdivo
View Photos of Packaging, Labels & Appearance
- image 05
- opdivo-100mg-10ml-pdp - opdivo 100mg 10ml pdp
- opdivo 120mg 12ml pdp
- opdivo-240mg-24ml-pdp - opdivo 240mg 24ml pdp
- opdivo-40mg-4ml-pdp - opdivo 40mg 4ml pdp
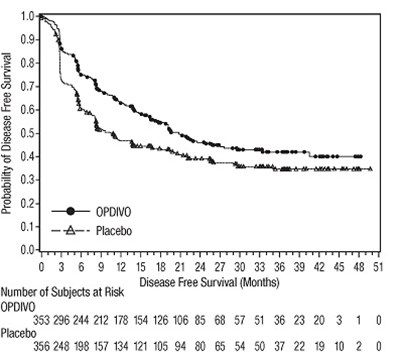
- opdivo dfs checkmate 274
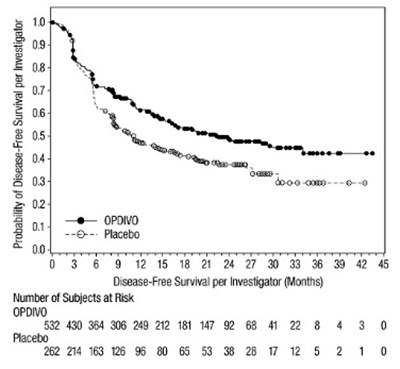
- opdivo-dfs-checkmate-577.jpg - opdivo dfs checkmate 577
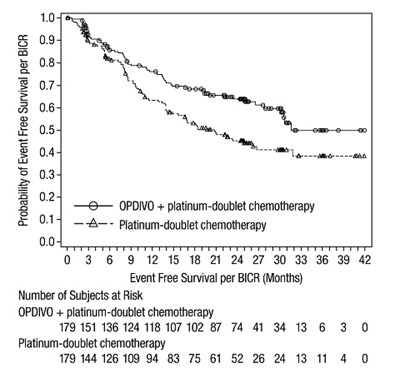
- opdivo efs checkmate 816
- opdivo-forest-os-checkmate-057.jpg - opdivo forest os checkmate 057
- opdivo-forest-pfs-checkmate-057.jpg - opdivo forest pfs checkmate 057
- opdivo-os-all-patients-checkmate-649.jpg - opdivo os all patients checkmate 649
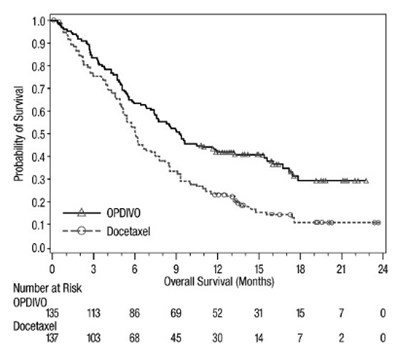
- opdivo-os-checkmate-017.jpg - opdivo os checkmate 017
- opdivo-os-checkmate-025.jpg - opdivo os checkmate 025
- opdivo-os-checkmate-037 - opdivo os checkmate 037
- opdivo-os-checkmate-057.jpg - opdivo os checkmate 057
- opdivo-os-checkmate-066.jpg - opdivo os checkmate 066
- opdivo-os-checkmate-067 - opdivo os checkmate 067
- opdivo-os-checkmate-141.jpg - opdivo os checkmate 141
- opdivo-os-checkmate-214.jpg - opdivo os checkmate 214
- opdivo-os-checkmate-227 - opdivo os checkmate 227
- opdivo os checkmate 648 A
- opdivo os checkmate 648 B
- opdivo-os-checkmate-743.jpg - opdivo os checkmate 743
- opdivo-os-checkmate-9er.jpg - opdivo os checkmate 9er
- opdivo-os-checkmate-9la.jpg - opdivo os checkmate 9la
- opdivo-os-pd-l1-cps-1-or-greater-checkmate-649.jpg - opdivo os pd l1 cps 1 or greater checkmate 649
- opdivo-os-pd-l1-cps-5-or-greater-checkmate-649.jpg - opdivo os pd l1 cps 5 or greater checkmate 649
- opdivo pfs checkmate 648 A
- opdivo pfs checkmate 648 B
- opdivo-pfs-checkmate-9er.jpg - opdivo pfs checkmate 9er
- opdivo-rfs-checkmate-238 - opdivo rfs checkmate 238
Product Label Images
The following 31 images provide visual information about the product associated with Opdivo NDC 0003-3734 by E.r. Squibb & Sons, L.l.c., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
image 05
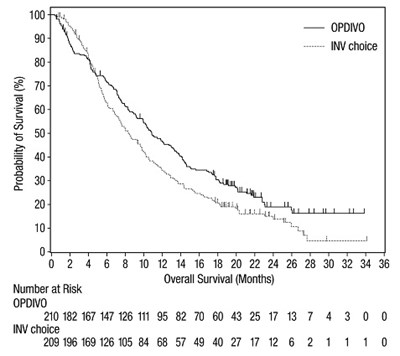
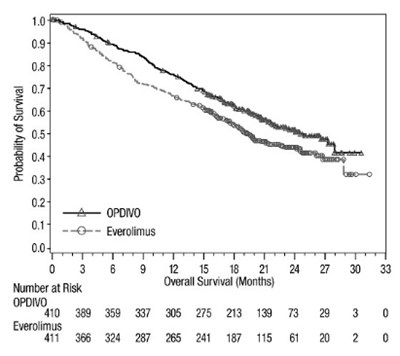
The provided text shows a graph with probability of survival on the y-axis and overall survival in months on the x-axis. However, some of the text is not properly recognized due to the error.*
opdivo-100mg-10ml-pdp - opdivo 100mg 10ml pdp
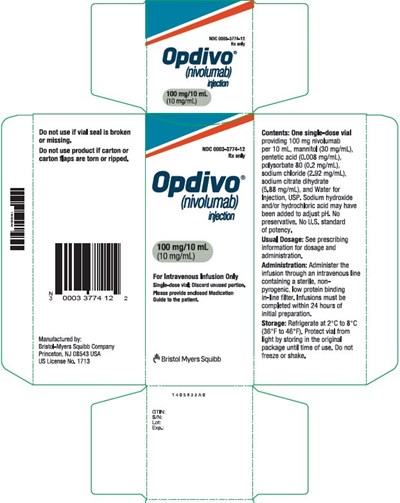
This is a warning label for an intravenous medication called "nvokimad". It cautions users not to use if the bottle, carton, or seal is damaged, and to only use it as directed by a physician. The medication contains 100mg of nvokimad per 10 mL of solution, along with other ingredients. The label outlines storage instructions for the medication, such as refrigerating it between uses and avoiding shaking or freezing the bottle.*
opdivo 120mg 12ml pdp

This is a description for a medication provided in a single dose vial. The product should not be used if the vial seal is broken or missing, or if the carton or carton flaps are torn or ripped. The medication is produced by Bristol Myers Squibb and is for intravenous infusion only. It contains 120 mg of the drug in 12 mL of solution and is administered through an intravenous line. The medication includes several other ingredients to adjust its pH and maintain its stability. The medication must be infused within 24 hours of preparation, and it should be refrigerated between 2-5°C until use. The dosage and administration information is provided in the prescribing information.*
opdivo-240mg-24ml-pdp - opdivo 240mg 24ml pdp

This is a description of a pharmaceutical product made by Bristol Myers Squibb CO. It contains a single-dose vial that is only for intravenous infusion. The product should not be used if the carton or carton flaps are torn or ripped. The ingredients include nivolumab, pentetic acid, polysorbate 80, sodium chloride, sodium citrate dihydrate, and water for injection. The administration should be through an intravenous non-pyrogenic low-potency binding-filtration device and completed within 24 hours of preparation. The product should be stored in a refrigerator at 2°C-5°C and protected from light in its original package.*
opdivo-40mg-4ml-pdp - opdivo 40mg 4ml pdp
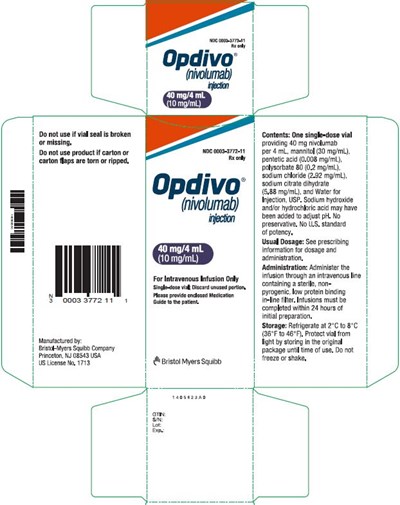
This is a medication warning label for a 40 mg/4 ml vial of Nivolumab. It is for intravenous infusion only. The label warns against using the product if the carton or carton flaps are torn, missing, or ripped. The medication should be discarded after use, and it should be protected from light. Dosage information is provided in the prescribing information, and the medication should be administered through a non-pyrogenic, low protein binding inline filter. The medication should be stored at 2°C to 8°C and should not be frozen or shaken.*
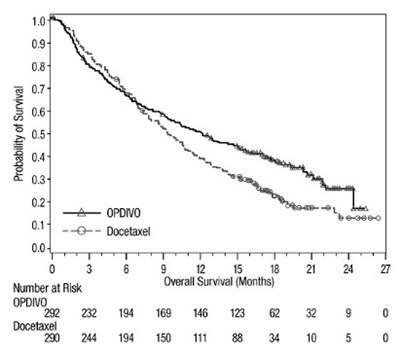
opdivo-forest-os-checkmate-057.jpg - opdivo forest os checkmate 057
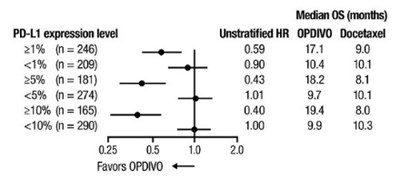
This appears to be a statistical table or chart related to treatment outcomes with two drugs OPDIVO and Docetaxel. It includes information about PD-L1 expression and median time periods in months. However, due to the lack of context and clear readability, a detailed description cannot be provided.*
opdivo-forest-pfs-checkmate-057.jpg - opdivo forest pfs checkmate 057

This is a study showing the median PFS (progression-free survival) in months for patients with PD-L1 expression level of 21% treated with OPDIVO and Docetaxel. The unstratified HR (hazard ratio) is also included in the table. However, no additional information or context is given, so it is unclear what the study is about or what the results mean.*
opdivo-os-all-patients-checkmate-649.jpg - opdivo os all patients checkmate 649

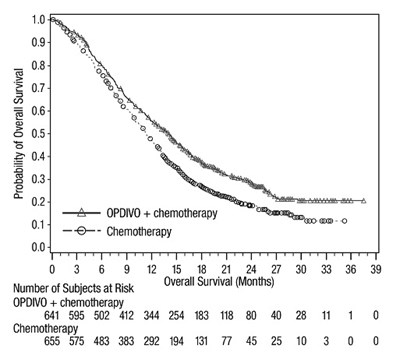
This appears to be a graph showing the probability of overall survival for subjects receiving OPDIVO in addition to chemotherapy versus those receiving only chemotherapy. The X-axis represents the number of subjects at specific time intervals and the Y-axis represents the duration of overall survival in months. The graph shows that the probability of overall survival is higher for subjects receiving OPDIVO in addition to chemotherapy compared to those receiving only chemotherapy. However, the data is truncated and incomplete as there is not enough information provided to interpret the graph accurately.*
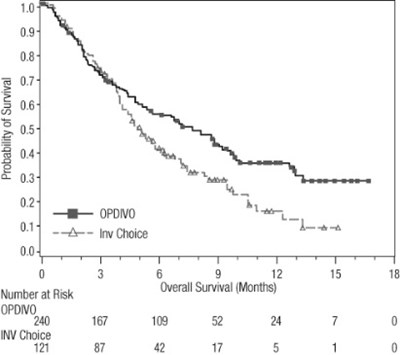
opdivo-os-checkmate-037 - opdivo os checkmate 037
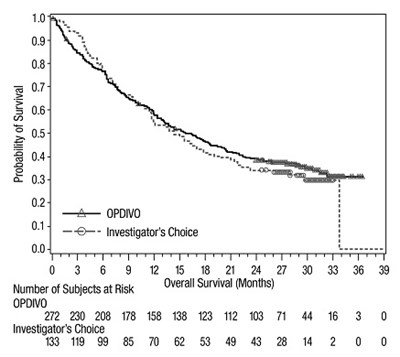
This appears to be a table displaying the Probability of Survival for a particular study or clinical trial. The dates or years may be represented in the first column, with corresponding survival rates in the subsequent columns. There is a section header labeled "Investigator's Choice" which may refer to a specific treatment group or intervention. The last row contains numerical values for the number of subjects at risk and their overall survival, measured in months. The origin or context of the table is not clear.*
opdivo-os-checkmate-066.jpg - opdivo os checkmate 066

This is a chart that shows the probability of survival over a certain number of months for two different treatments: one with the drug Dacarbazine and another treatment labeled as TR. The chart has numbered intervals on the horizontal axis and months on the vertical axis. The TR treatment appears to have a slightly higher probability of survival at the beginning, but the Dacarbazine treatment becomes more effective after three months.*
opdivo-os-checkmate-067 - opdivo os checkmate 067
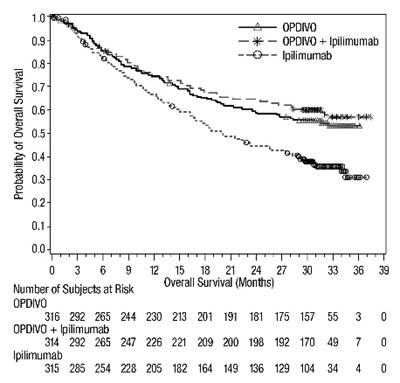
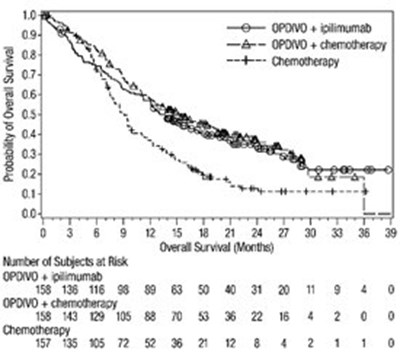
This appears to be a table that compares the survival rates of several subjects using different treatments. The treatments being compared are OPDIVO, OPDIVO + Ipiimumab, and Iplimumab. The table shows the number of subjects at different overall survival months.*
opdivo-os-checkmate-214.jpg - opdivo os checkmate 214
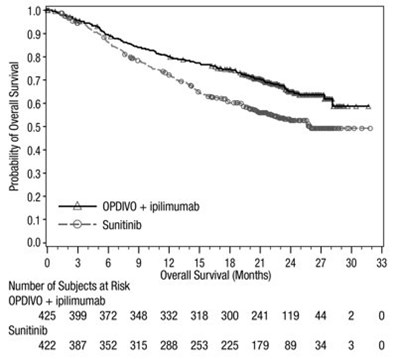
The generated text is a chart depicting the overall survival rates in months for two treatment options, OPDIVO + ipilimumab and Sunitinib. The chart also shows the number of subjects at risk for each treatment option.*
opdivo-os-checkmate-227 - opdivo os checkmate 227
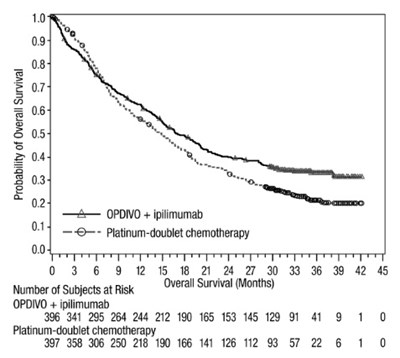
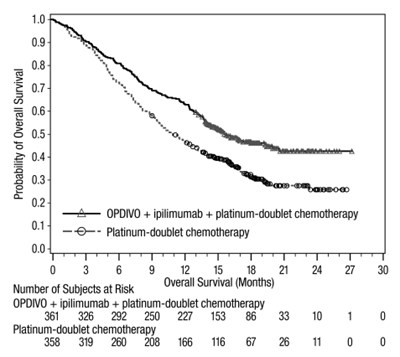
This is a graph showing the probability of overall survival for two groups of subjects, one treated with OPDVO and ipilimumab and the other group with platinum-doublet chemotherapy. The X-axis represents the months elapsed since the start of treatment and the Y-axis represents the number of subjects who survived.*
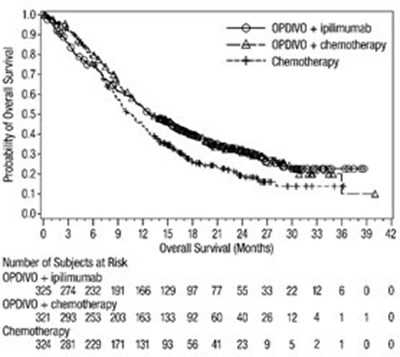
opdivo-os-checkmate-743.jpg - opdivo os checkmate 743

This text provides data on the probability of overall survival in subjects treated with OPDIVO+ipilimumab and chemotherapy. The data includes the number of subjects at risk, overall survival (in months), and the treatment method used.*
opdivo-pfs-checkmate-9er.jpg - opdivo pfs checkmate 9er
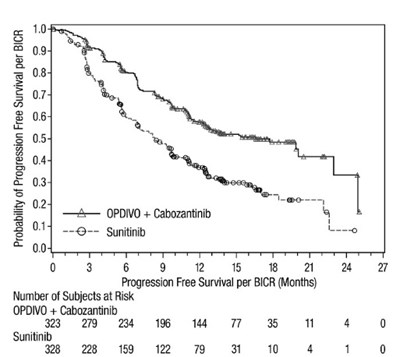
The text provides a chart displaying the probability of progression-free survival per BICR for two treatments (OPDIVO + Cabozantinib and Sunitinib) over a period of months. The number of subjects at risk is also shown for each treatment. No further information is available.*
opdivo-rfs-checkmate-238 - opdivo rfs checkmate 238
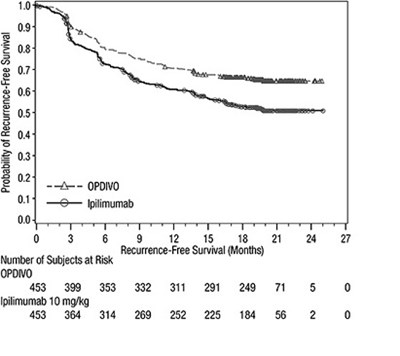
This appears to be a survival analysis table showing the probability of recurrence-free survival for subjects receiving treatment with ipilimumab. The table also includes the number of subjects at risk at each time point.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.