Product Images Keytruda
View Photos of Packaging, Labels & Appearance
- Figure 1 - keytruda 01
- Figure 2 - keytruda 02
- Figure 3 - keytruda 02a
- Figure 4 - keytruda 03
- Figure 7 - keytruda 04
- Figure 5 - keytruda 04a
- Figure 6 - keytruda 04b
- Figure 8 - keytruda 05
- Figure 9 - keytruda 05a
- Figure 10 - keytruda 05b
- Figure 11 - keytruda 05c
- Figure 12 - keytruda 06
- Figure 15 - keytruda 06a
- Figure 16 - keytruda 06b
- Figure 13 - keytruda 06c
- Figure 17 - keytruda 06d
- Figure 14 - keytruda 06e
- keytruda 06f
- keytruda 06g
- keytruda 06h
- keytruda 06i
- keytruda 06j
- keytruda 06k
- keytruda 06l
- keytruda 06m
- keytruda 06n
- PRINCIPAL DISPLAY PANEL - 50 mg Vial Carton - keytruda 07
- PRINCIPAL DISPLAY PANEL - 100 mg/4 mL Vial Carton - keytruda 08
Product Label Images
The following 28 images provide visual information about the product associated with Keytruda NDC 0006-3029 by Merck Sharp & Dohme Llc, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Figure 2 - keytruda 02

This appears to be a chart or graph showing progression-free survival rates in months for different treatments. The treatments mentioned are "KEYTRUDA 1mg" and "KEYTRUDA 2mg", as well as "Cremaneragy". The y-axis represents the percentage of progression-free survival, while the x-axis represents time in months. The percentage values range from 0 to 30, and the time values range from 0 to 21 months. There is also a "Number at Risk" column, but it is unclear what this refers to without further context.*
Figure 6 - keytruda 04b

This appears to be a table or graph displaying overall survival percentages for a treatment arm (listed as "KEYTRUDA") and chemotherapy over time in months. The table also includes the number at risk for each treatment option. However, the information is not detailed enough to provide a specific description or interpretation without additional context.*
Figure 15 - keytruda 06a

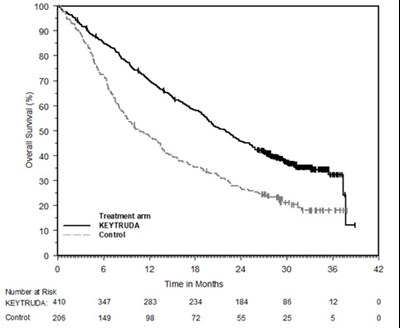
This is a table or chart showing the percentage of overall survival in a treatment arm (Keytruda) and a control group. It also lists the number of individuals at risk and the time in months. There are some unreadable characters and numbers.*
keytruda 06h

This is a graph showing the event-free survival of patients treated with KEYTRUDA and chemotherapy compared to patients treated with a placebo and chemotherapy. The graph shows the percent of patients who have not experienced an event related to the disease or treatment over time. The numbers on the bottom indicate the number of patients at risk and the time in months.*
keytruda 06k

This appears to be a table displaying the efficacy of the KEYTRUDA Chematherspy treatment arm compared to the effectiveness of chemotherapy, with percentages ranging from 0% to 100% overall Sunival. The table also displays the percentage of treatment efficacy at different points in time (in months). It is unclear what "Number st Risk" refers to.*
keytruda 06n
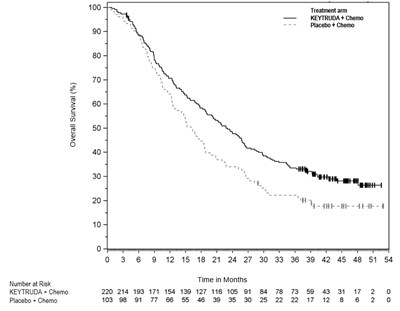
This appears to be a chart or graph displaying data points for two different treatments, KEVTRUDA + Chemo and Placebo + Chemo, over a period of 54 months. The X-axis represents time in months and the Y-axis represents the number of patients responding to treatment. However, it is difficult to determine the exact nature of the treatment, the response being measured, or the source of the data.*
PRINCIPAL DISPLAY PANEL - 50 mg Vial Carton - keytruda 07

This appears to be a description of Keytruda, a medication used for intravenous infusion only. It contains the active ingredient pembrolizumab and is manufactured and distributed by Merck Sharp & Dohme Corp. The dosage instructions and storage information are not provided, but it appears to come in vials of 50mg. The text also includes copyright and legal information related to the medication. No further information is available due to the limited amount of text.*
PRINCIPAL DISPLAY PANEL - 100 mg/4 mL Vial Carton - keytruda 08

Keytruda® is a medication used for intravenous infusion only. Each vial contains 100mg of pembrolizumab in 4 mL of solution, with each 1 mL of solution containing 25 mg pembrolizumab, Lohistidine (15 mg), polysorbate 80 (0.1 mg), sucrose (70mg), and Water for Injection, USP with no preservative. The medication must be refrigerated at 2°C-8°C (6°F- 46°F) in its original packaging to prevent light exposure, and it should not be frozen or shaken. The typical dosage is documented on the package insert, and any unused portion must be discarded due to its single-dose vial. A medication guide is enclosed for each patient, and dilution is required before administration. The manufacturer is a subsidiary of Merck and Co., and the product is manufactured in Germany.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.