Product Images Renflexis
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 5 images provide visual information about the product associated with Renflexis NDC 0006-4305 by Merck Sharp & Dohme Llc, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Figure 3 - renflexis 03
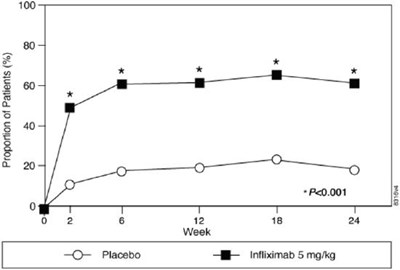
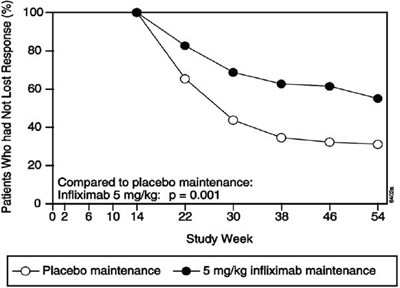
This text appears to describe two different medical interventions: "Placebo" and "Infliximab 5 mg/kg". Beyond that, there is not enough context to determine what conditions the interventions might be used for or what their respective effects might be.*
PRINCIPAL DISPLAY PANEL - 100 mg Vial Carton - renflexis 05

This is a description of RENFLEXIS™ medication for injection. It is available in 100mg/vial dosage. The medication must be refrigerated at a temperature of 2-8°C and has a shelf life of up to 6 months. The product must be reconstituted and diluted before intravenous infusion, and must be infused over at least 2 hours with an inline filter. The instructions for preparing the medication are provided on the package. It is manufactured by Samsung Bioepis and distributed by Merck Sharp & Dohme. It is meant for intravenous infusion only and must be administered by a healthcare professional. There is a packaging insert with medication guide that should be given to each patient. The medication contains Infliximab-abda. No usage information is available if the text contains it.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.