Product Images Duavee
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 6 images provide visual information about the product associated with Duavee NDC 0008-1123 by Wyeth Pharmaceuticals Llc, A Subsidiary Of Pfizer Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Logo - duavee 02

This is a statement indicating that a company called Wyeth Pharmaceuticals LLC is being distributed by another company called Pfizer Inc, and is based in Philadelphia, with a zip code of 19101.*
Logo - duavee 02a

This text is a statement that indicates the distributor of the product which is Wyeth Pharmaceuticals LLC. It is a subsidiary of Pizer Inc in Philadelphia, PA 19101.*
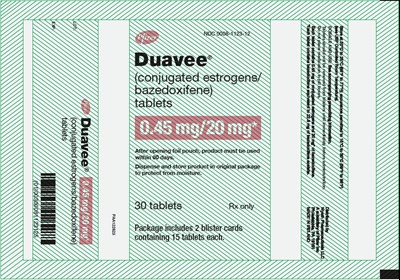
PRINCIPAL DISPLAY PANEL - 30 Tablet Blister Pack Pouch Carton - duavee 05
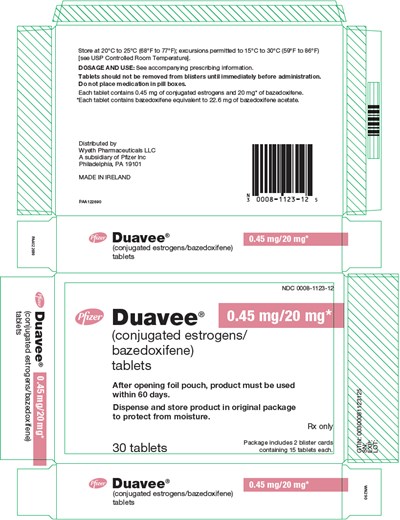
Duavee® (conjugated estrogens/bazedoxifene) is a medication that comes in a pack of 30 tablets. Each tablet contains 0.45 mg of conjugated estrogens and 20 mg of bazedoxifene. The tablets should not be removed from the blisters until immediately before administration and shouldn't be placed in pill boxes. The recommended storage temperature is 20-25°C with permitted excursions of 15-30°C. The product must be used within 60 days of opening the foil pouch, and it should be kept in the original package to protect from moisture. See the accompanying prescribing information for dosage and use. Distributed by Wyeth Pharmaceuticals, a subsidiary of Pfizer Inc. Made in Ireland.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.