Product Images Genotropin
View Photos of Packaging, Labels & Appearance
- Logo - genotropin 01
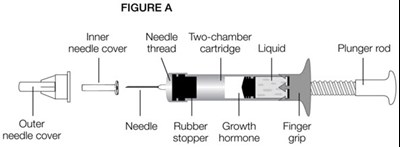
- Figure A - genotropin 02
- Figure B - genotropin 03
- Figure C - genotropin 04
- Figure D - genotropin 05
- Figure E - genotropin 06
- Figure F - genotropin 07
- Figure G - genotropin 08
- Figure H - genotropin 09
- Figure I - genotropin 10
- Principal Display Panel - 0.4 mg Cartridge Label - genotropin 100
- Principal Display Panel - 0.4 mg Kit Carton - genotropin 101
- Principal Display Panel - 0.6 mg Cartridge Label - genotropin 102
- Principal Display Panel - 0.6 mg Kit Carton - genotropin 103
- Principal Display Panel - 0.8 mg Cartridge Label - genotropin 104
- Principal Display Panel - 0.8 mg Kit Carton - genotropin 105
- Principal Display Panel - 1 mg Cartridge Label - genotropin 106
- Principal Display Panel - 1 mg Kit Carton - genotropin 107
- Principal Display Panel - 1.2 mg Cartridge Label - genotropin 108
- Principal Display Panel - 1.2 mg Kit Carton - genotropin 109
- Figure J - genotropin 11
- Principal Display Panel - 1.4 mg Cartridge Label - genotropin 110
- Principal Display Panel - 1.4 mg Kit Carton - genotropin 111
- Principal Display Panel - 1.6 mg Cartridge Label - genotropin 112
- Principal Display Panel - 1.6 mg Kit Carton - genotropin 113
- Principal Display Panel - 1.8 mg Cartridge Label - genotropin 114
- Principal Display Panel - 1.8 mg Kit Carton - genotropin 115
- Principal Display Panel - 2 mg Cartridge Label - genotropin 116
- Principal Display Panel - 2 mg Kit Carton - genotropin 117
- Logo - genotropin 12
- Logo - genotropin 13
- Logo - genotropin 14
- Figure A - genotropin 15
- Figure B - genotropin 16
- Figure C - genotropin 17
- Figure D - genotropin 18
- Figure E - genotropin 19
- Figure F - genotropin 20
- Figure G - genotropin 21
- Figure H - genotropin 22
- Figure I - genotropin 23
- Figure J - genotropin 24
- Figure K - genotropin 25
- Figure L - genotropin 26
- Figure M - genotropin 27
- Figure N - genotropin 28
- Figure O - genotropin 29
- Figure P - genotropin 30
- Image - genotropin 31
- Figure Q - genotropin 32
- Figure R - genotropin 33
- Figure S - genotropin 34
- Figure T - genotropin 35
- Figure U - genotropin 36
- Figure V - genotropin 37
- Figure W - genotropin 38
- Figure X - genotropin 39
- Figure Y - genotropin 40
- Figure Z - genotropin 41
- Figure AA - genotropin 42
- Image - genotropin 43
- Image - genotropin 44
- Image - genotropin 45
- Image - genotropin 46
- Image - genotropin 47
- Image - genotropin 48
- Image - genotropin 49
- Image - genotropin 50
- Image - genotropin 51
- Image - genotropin 52
- Logo - genotropin 53
- Logo - genotropin 54
- Figure A - genotropin 55
- Figure B - genotropin 56
- Figure C - genotropin 57
- Figure D - genotropin 58
- Figure E - genotropin 59
- Figure F - genotropin 60
- Figure G - genotropin 61
- Figure H - genotropin 62
- Figure I - genotropin 63
- Figure J - genotropin 64
- Figure K - genotropin 65
- Figure L - genotropin 66
- Figure M - genotropin 67
- Figure N - genotropin 68
- Figure O - genotropin 69
- Figure P - genotropin 70
- Image - genotropin 71
- Figure Q - genotropin 72
- Figure R - genotropin 73
- Figure S - genotropin 74
- Figure T - genotropin 75
- Figure U - genotropin 76
- Figure V - genotropin 77
- Figure W - genotropin 78
- Figure X - genotropin 79
- Figure Y - genotropin 80
- Figure Z - genotropin 81
- Figure AA - genotropin 82
Product Label Images
The following 100 images provide visual information about the product associated with Genotropin NDC 0013-2658 by Pfizer Laboratories Div Pfizer Inc, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Principal Display Panel - 0.4 mg Cartridge Label - genotropin 100

Genotropin® [EI NN] Esomalropin) for injection is a growth hormone delivery device manufactured by Pharmacia & Upjohn Co, now a subsidiary of Pfizer Inc. The specific product being referred to is the Genotropin MiniQuick® which has the NDC code 0013-2650-01. The Lot and EXP numbers are not available.*
Principal Display Panel - 0.6 mg Cartridge Label - genotropin 102

Genotropin® [N Esomalropin) is a growth hormone injection device produced by Pharmacia & Upjohn Co, now a part of Pfizer Inc. The device is called Genotropin MiniQuick® and has the NDC 0013-2651-01 code. The LOT and EXP are not available from the given text, and further information may be required.*
Principal Display Panel - 0.8 mg Cartridge Label - genotropin 104

Genotropin® is an injectable form of growth hormone delivery device manufactured by Pharmacia & Upjohn Co, a division of Pfizer Inc, NY. The device is called Genotropin MiniQuick® and has an NDC number. The provided text doesn't contain any information about the expiration date or lot number.*
Principal Display Panel - 0.8 mg Kit Carton - genotropin 105

Genotropin® (somatropin) for Injection is a medicine that is available in the form of 0.8mg Genotropin MiniQuick for single subcutaneous injection. It is a Growth Hormone Delivery Device that comes in a package of 7. It is meant to be used for medical purposes only and is a prescription drug.*
Principal Display Panel - 1 mg Cartridge Label - genotropin 106

Genotropin® is a growth hormone delivery device with the trade name Esomatropln. The device is manufactured by Pharmacia & Upjohn Co, a division of Pfizer Inc. The specific product is Genotropin MiniQuick® and is identified by NDC 0013-2653-01. The provided code PAA142733 is likely associated with the product's quality control. There is no additional information available.*
Principal Display Panel - 1 mg Kit Carton - genotropin 107

This is a description of a medication called "Genotropin" used for growth hormone delivery via a single subcutaneous injection, and comes in a pack of seven. The text includes various alphanumeric codes that appear to be identification numbers or lot numbers. There is also some text that appears to be unrelated and is not readable.*
Principal Display Panel - 1.2 mg Cartridge Label - genotropin 108

Genotropin is a brand name for somatropin injection. The text also mentions Enatropin MiniQuick as a growth hormone delivery system by Pharmacia & Upjohn Co, a division of Pfizer Inc. The text contains some unclear characters and random special characters, however, the critical information about the product name and manufacturer is understandable.*
Principal Display Panel - 1.2 mg Kit Carton - genotropin 109

Genotropin® is a medication for single subcutaneous injection only and is used for Growth Hormone Delivery Device. The NDC for this medication is 0012-2654-02 and it is intended for prescription use only. The majority of the text provided is not readable or non-English characters.*
Principal Display Panel - 1.4 mg Kit Carton - genotropin 111

This is a product description of the Genotropin® Growth Hormone Delivery Device in the form of a 1.4 mg Genotropin MiniQuick® for single subcutaneous injection only. The product is available in a pack of 7. The text seems to be surrounded by unrelated characters and gibberish.*
Principal Display Panel - 1.6 mg Cartridge Label - genotropin 112

Genotropin® is a brand name for somatropin, which is a type of growth hormone used for injection. Genotropin MiniQuick® is a delivery device for this hormone. The product is manufactured by Pharmacia & Upjohn Co, which is a subsidiary of Pfizer Inc, based in New York. The provided text doesn't include information about the lot or expiration date.*
Principal Display Panel - 1.6 mg Kit Carton - genotropin 113

The text provided is a product description of Genotropin® - a brand of injectable human growth hormone. It specifies the dosage, delivery method, and packaging for the product. It also indicates that it is for the single subcutaneous injection only and should only be used under a doctor's prescription.*
Principal Display Panel - 1.8 mg Cartridge Label - genotropin 114

Genotropin® EETT I (somatropin) for injection is a growth hormone medication that comes in a delivery device called Genotropin MiniQuick®. The medication is distributed by the Pharmacia & Upjohn Co, which is a division of Pfizer Inc located in New York City. The text also includes a lot number and expiration date, but no further information is available.*
Principal Display Panel - 2 mg Cartridge Label - genotropin 116

Genotropin® P I is a growth hormone delivery device known as Esomalropin for injection. It includes a mini-quick device with NDC 0013-2658-01 to deliver the hormone. The product is made by Pharmacia & Upjohn Company, which is now a part of Pfizer Inc, located in New York, NY. The text also includes a LOT and EXP number, which is likely a batch or lot number and expiration date related to the product.*
Principal Display Panel - 2 mg Kit Carton - genotropin 117

Genotropin® (somatropin) is a prescription medicine used for injection for growth hormone delivery. The 2 Mg Genotropin MiniQuick® is suitable for single subcutaneous administration only. The package contains 7 Genotropin® growth hormone delivery devices. The NDC number for the package is 0012-2658.02.*
Logo - genotropin 12

This is a description of a pharmaceutical product which is distributed by Pharmacia & Upjohn Co, a division of Pfizer Inc, located in NY, NY 10017.*
Logo - genotropin 14

The text describes a growth hormone delivery device called Genotropin ren°12, which is designed to be used with an injectable medication called Genotropin (somatropin).*
Figure A - genotropin 15

Metal Indicative Dose Injection is a medical device that involves the injection of medicine into the body. The device comes with a needle guard, outer layer needle, and a cartridge. The front cap of the device is red in color and is used for cleaning purposes after injection. The rubber stopper is also part of the cartridge.*
Logo - genotropin 53

This is a description of a distributor named Pharmacia & Upjohn Co, which is a division of Pfizer Inc located in NY, NY 10017.*
Logo - genotropin 54

Genotropin pen°5 is a delivery device intended for use with the medication Genotropin (somotropin) for injection. This device is used to administer growth hormone.*
Figure A - genotropin 55
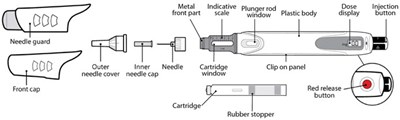
This text appears to be a diagram or parts list for a needle guard apparatus, likely for a medical injection device. It includes labels for various parts such as the outer and inner covers for the needle, a front cap, metal front part, plunger, dose scale, plastic body, and cartridge clip-on panel. The cartridge itself is described as having a window, a rubber stopper, a red release button, and an injection button. Overall, it seems to be a technical description of a specific device rather than a general product description.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.