Product Images Durysta
View Photos of Packaging, Labels & Appearance
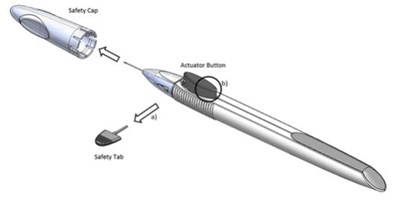
- Figure 1 - durysta 01
- Figure 2 - durysta 02
- The structural formula for DURYSTA is an intracameral implant containing 10 mcg bimatoprost in the NOVADUR® solid polymer sustained-release drug delivery system (DDS). DURYSTA is preloaded into a single-use, DDS applicator to facilitate injection of the rod-shaped implant directly into the anterior chamber of the eye. The components of the implant are bimatoprost drug substance and polymers poly (D,L-lactide), poly (D,L-lactide-co-glycolide), and polyethylene glycol. The chemical name for bimatoprost is (Z)-7-[(1R,2R,3R,5S)-3,5-dihydroxy-2-[(1E,3S)-3-hydroxy-5-phenyl-1-pentenyl]cyclopentyl]-N-ethyl-5-heptenamide, and its molecular weight is 415.57. Its molecular formula is C25H37NO4. - durysta 03
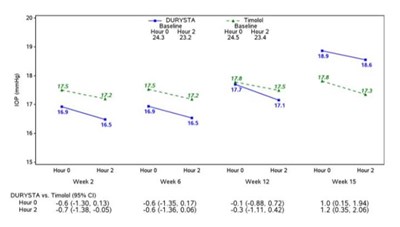
- Figure 3: Study 1 Mean IOP (mmHg) by Treatment Group and Treatment Difference in Mean IOP - durysta 04
- Figure 4: Study 2 Mean IOP (mmHg) by Treatment Group and Treatment Difference in Mean IOP - durysta 05
- PRINCIPAL DISPLAY PANELDURYSTATM(bimatoprost implant) 10 mcgNDC 0023-9652-01 - durysta 07
Product Label Images
The following 6 images provide visual information about the product associated with Durysta NDC 0023-9652 by Allergan, Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
The structural formula for DURYSTA is an intracameral implant containing 10 mcg bimatoprost in the NOVADUR® solid polymer sustained-release drug delivery system (DDS). DURYSTA is preloaded into a single-use, DDS applicator to facilitate injection of the rod-shaped implant directly into the anterior chamber of the eye. The components of the implant are bimatoprost drug substance and polymers poly (D,L-lactide), poly (D,L-lactide-co-glycolide), and polyethylene glycol. The chemical name for bimatoprost is (Z)-7-[(1R,2R,3R,5S)-3,5-dihydroxy-2-[(1E,3S)-3-hydroxy-5-phenyl-1-pentenyl]cyclopentyl]-N-ethyl-5-heptenamide, and its molecular weight is 415.57. Its molecular formula is C25H37NO4. - durysta 03
![durysta-03.jpg The structural formula for DURYSTA is an intracameral implant containing 10 mcg bimatoprost in the NOVADUR® solid polymer sustained-release drug delivery system (DDS). DURYSTA is preloaded into a single-use, DDS applicator to facilitate injection of the rod-shaped implant directly into the anterior chamber of the eye. The components of the implant are bimatoprost drug substance and polymers poly (D,L-lactide), poly (D,L-lactide-co-glycolide), and polyethylene glycol. The chemical name for bimatoprost is (Z)-7-[(1R,2R,3R,5S)-3,5-dihydroxy-2-[(1E,3S)-3-hydroxy-5-phenyl-1-pentenyl]cyclopentyl]-N-ethyl-5-heptenamide, and its molecular weight is 415.57. Its molecular formula is C25H37NO4. - durysta 03](https://ndclist.com/assets/spl/images/3f59da84-0bcc-4c84-b3e2-e215681ef341/400x-durysta-03.jpg)
Figure 3: Study 1 Mean IOP (mmHg) by Treatment Group and Treatment Difference in Mean IOP - durysta 04