Product Images Plavix
View Photos of Packaging, Labels & Appearance
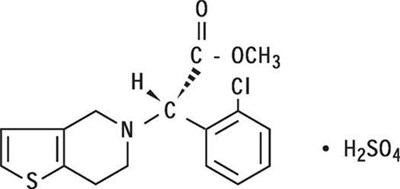
- Chemical Structure - plavix 01
- Figure - plavix 02
- Figure - plavix 03
- Figure - plavix 04
- Figure - plavix 05
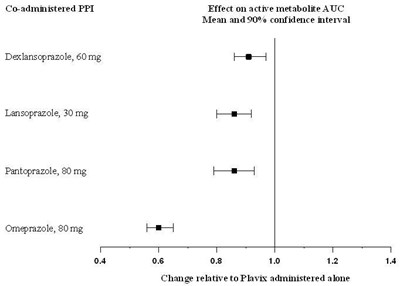
- Figure - plavix 06
- Figure - plavix 07
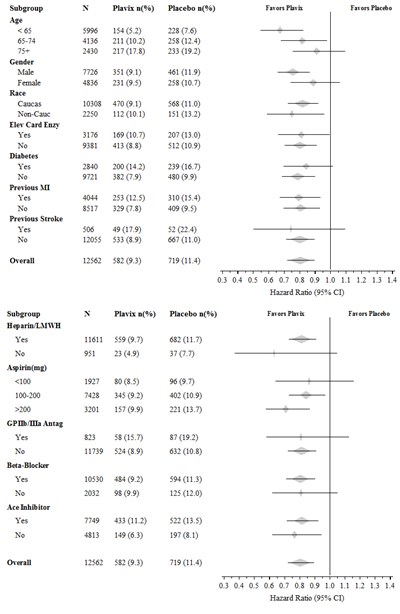
- Figure - plavix 08
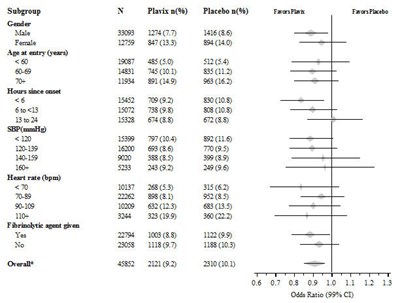
- Figure - plavix 09
- PRINCIPAL DISPLAY PANEL - 75 mg Tablet Bottle Label - plavix 10
- PRINCIPAL DISPLAY PANEL - 300 mg Tablet Blister Pack Carton - plavix 11
Product Label Images
The following 11 images provide visual information about the product associated with Plavix NDC 0024-1332 by Sanofi-aventis U.s. Llc, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
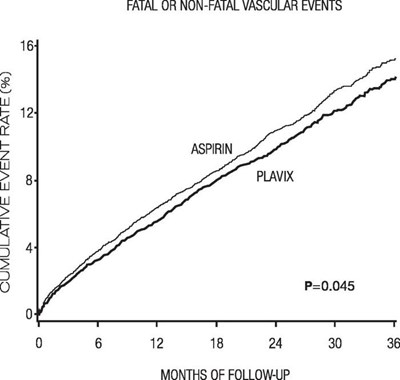
Figure - plavix 03
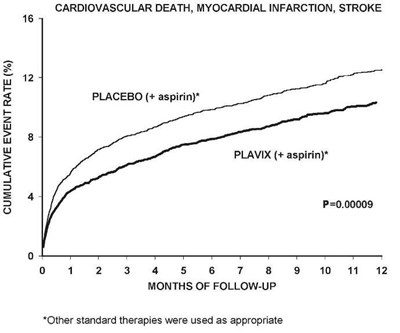
The text describes a chart showing the cumulative event rate for cardiovascular death, myocardial infarction, and stroke in patients taking either a placebo with aspirin or Plavix with aspirin. The chart indicates that Plavix with aspirin has a significantly lower cumulative event rate compared to the placebo with aspirin. The chart covers a follow-up period of up to 12 months and notes that other standard therapies were used as appropriate.*
Figure - plavix 05
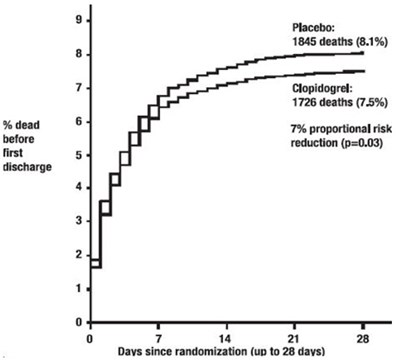
The given text compares the percentage of deaths between two groups - Placebo and Clopidogrel. The placebo group had 1845 deaths (8.1%) while the Clopidogrel group had 1726 deaths (7.5%). There was a 7% risk reduction in the Clopidogrel group (p=0.03). The text also includes a graph showing the number of days since randomization, which ranges up to 28 days.*
Figure - plavix 06
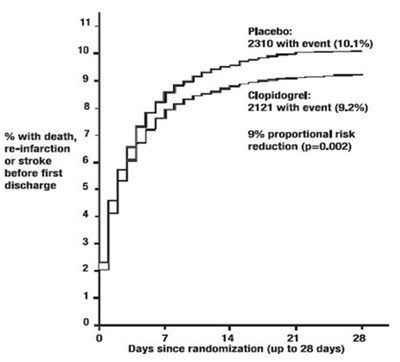
This is a statistical analysis of the effectiveness of Placebo compared to Clopidogrel in reducing the risk of death, re-infarction, or stroke before the first discharge. The data suggests that Clopidogrel shows a 9% proportional reduction in risk compared to Placebo. The percentage of people with death was reduced by 7%. The analysis was conducted up to 28 days since randomization.*
Figure - plavix 08
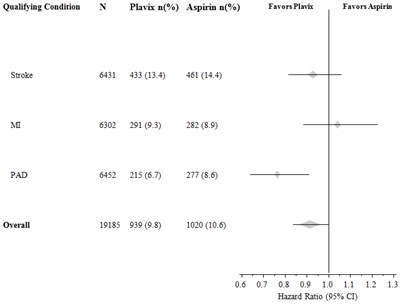
This text contains the results of a study comparing the effectiveness of Plavix and Aspirin in patients with stroke or PAD. The table displays the number of patients with qualifying conditions and the number receiving each medication, as well as the percentage for each group. The last line shows a hazard ratio with a confidence interval.*
PRINCIPAL DISPLAY PANEL - 75 mg Tablet Bottle Label - plavix 10

Clopidogrel tablets are a prescription medication manufactured by Sanofi. Each tablet contains 97.875 mg of clopidogrel sulfate equivalent to 75 mg of clopidogrel base. This medication is dispensed with a medication guide and the usual adult dosage can be found in the package insert. Plavix® is a registered trademark associated with this medication. For inquiries about the medication, individuals can call 1-800-446-6267. Store this medication at 25°C (77°F), with excursions permitted between 15°C-30°C (58°F-86°F) in accordance with USP Controlled Room Temperature guidelines.*
PRINCIPAL DISPLAY PANEL - 300 mg Tablet Blister Pack Carton - plavix 11

This is a description of a medication called Plavix manufactured by Sanofi in the form of 30 film-coated tablets containing 391.5 mg of clopidogrel bisulfate equivalent to 300 mg of clopidogrel base. The usual dosage is available in the package insert, and the medication is stored at a temperature of 25°C (77°F) with excursions between 15°C-30°C (59°F-86°F). The product is manufactured by Sanofiaventis U.S, LLC in Bridgewater, NJ 08807, and Plavix® is a registered trademark which can be inquired about at 1-800-633-1610. The text did not provide clear details regarding the indications or purpose for the medication.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.