Product Images Soliqua 100/33
View Photos of Packaging, Labels & Appearance
- Chemical Structure - soliqua 01
- Chemical Structure - soliqua 02
- image - soliqua 03
- image - soliqua 04
- image - soliqua 05
- image - soliqua 06
- image - soliqua 07
- image - soliqua 08
- image - soliqua 09
- image - soliqua 10
- image - soliqua 11
- image - soliqua 12
- image - soliqua 13
- image - soliqua 14
- image - soliqua 15
- image - soliqua 16
- image - soliqua 17
- image - soliqua 18
- image - soliqua 19
- image - soliqua 20
- image - soliqua 21
- image - soliqua 22
- image - soliqua 23
- image - soliqua 24
- image - soliqua 25
Product Label Images
The following 25 images provide visual information about the product associated with Soliqua 100/33 NDC 0024-5761 by Sanofi-aventis U.s. Llc, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Chemical Structure - soliqua 02
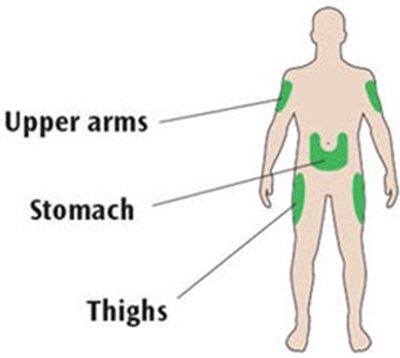
This is a list of body parts including upper arms, stomach and thighs. It does not provide any further context or information.*
image - soliqua 03
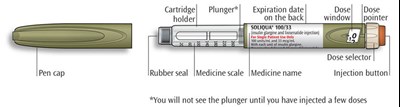
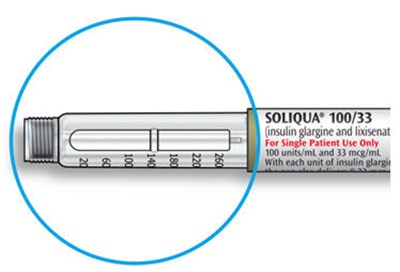
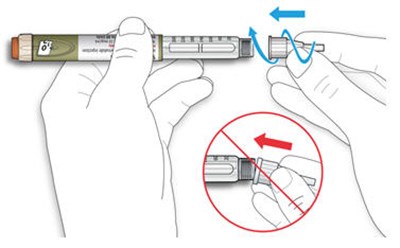
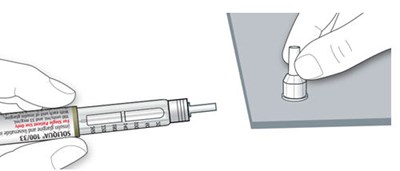
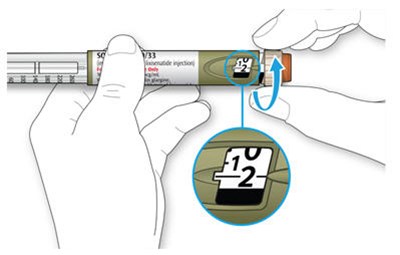
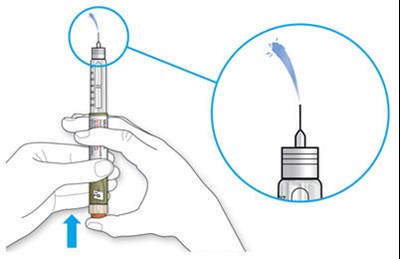
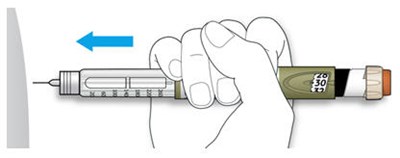
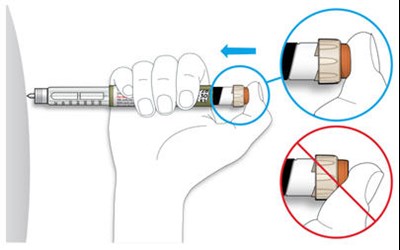
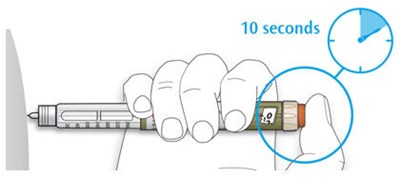
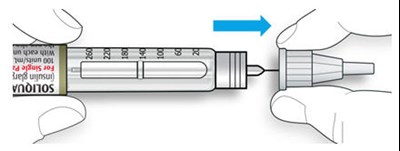
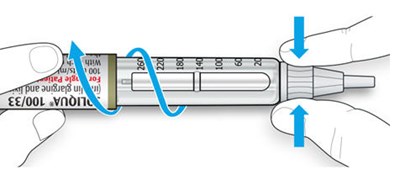
The text describes the design of a medication injection device. It includes a dose selector, rubber seal, medicine scale, medicine name, injection button, and a note that the plunger will not be visible until a few doses have been administered. The text also mentions the expiration date being located on the back.*
image - soliqua 25

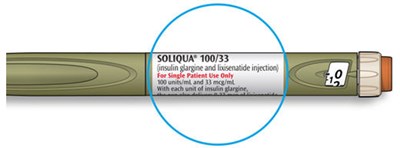
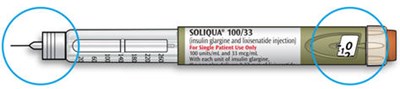
SOLIQUA® 100/33 is a prescription injection medication consisting of 100 units/mL insulin glargine and 33 mcg/mL lixisenatide. It is intended solely for subcutaneous injections and for single patient use only. SOLIQUA® 100/33 should be refrigerated at 36°F to 46°F (2C to 8°C) prior to first use, and protected from light. After first use, the pen should be stored at room temperature up to 77°F (25°C) for 28 days before disposal. A medication guide and secure sticker should be included by the supplier. Needles sold by separate manufacturers are compatible. Dosage adjustments should be made cautiously under medical supervision, and its use is not recommended for pregnant or nursing women without appropriate professional advice. Keep out of reach of children.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.