Product Images Toujeo
View Photos of Packaging, Labels & Appearance
- Chemical Structure - toujeo 01
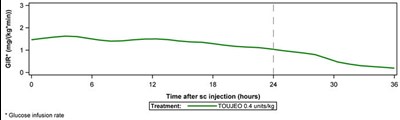
- Figure 1 - toujeo 02
- Figure - toujeo 03
- Figure - toujeo 04
- Figure - toujeo 05
- Figure - toujeo 06
- Figure - toujeo 07
- Figure - toujeo 08
- Figure - toujeo 09
- Figure - toujeo 10
- Figure - toujeo 11
- Figure - toujeo 12
- Figure - toujeo 13
- Figure - toujeo 14
- Figure - toujeo 15
- Figure - toujeo 16
- Figure - toujeo 17
- Figure - toujeo 18
- Figure - toujeo 19
- Figure - toujeo 20
- Figure - toujeo 21
- Figure - toujeo 22
- Figure - toujeo 23
- Figure - toujeo 24
- Figure - toujeo 25
- figure - toujeo 26
- Figure - toujeo 27
- Figure - toujeo 28
- Figure - toujeo 29
- Figure - toujeo 30
- Figure - toujeo 31
- Figure - toujeo 32
- Figure - toujeo 33
- Figure - toujeo 34
- Figure - toujeo 35
- Figure - toujeo 36
- Figure - toujeo 37
- Figure - toujeo 38
- Figure - toujeo 39
- Figure - toujeo 40
- Figure - toujeo 41
- Figure - toujeo 42
- Figure - toujeo 43
- Figure - toujeo 44
- Figure - toujeo 45
- Figure - toujeo 46
- Figure - toujeo 47
- Figure - toujeo 48
- Figure - toujeo 49
- Figure - toujeo 50
- PRINCIPAL DISPLAY PANEL - 1.5 mL Pen Carton - toujeo 51
- PRINCIPAL DISPLAY PANEL - 3 mL Pen Carton - toujeo 52
Product Label Images
The following 52 images provide visual information about the product associated with Toujeo NDC 0024-5869 by Sanofi-aventis U.s. Llc, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Figure - toujeo 03

This text indicates specific body parts that could be targeted during a workout or used to monitor changes in physical fitness. Upper arms, stomach, and thighs are commonly focused on during exercise as they are areas that people may want to tone or strengthen.*
Figure - toujeo 04

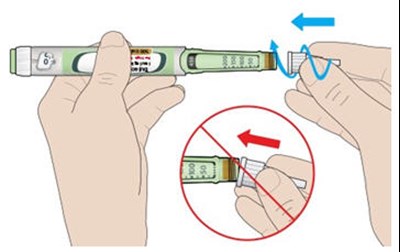
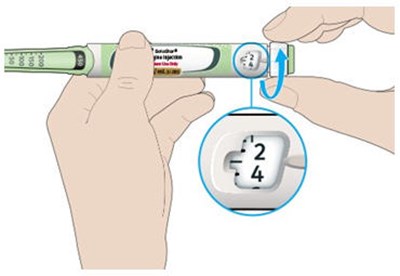
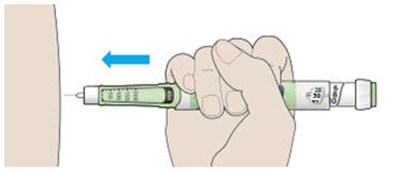
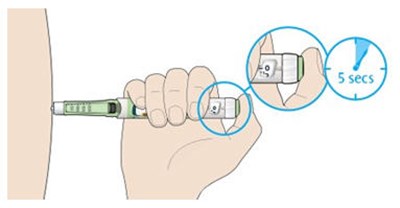
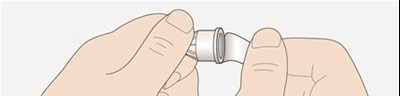
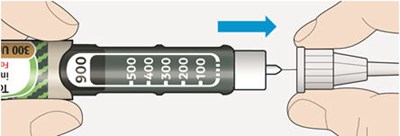
This appears to be a diagram or schematic of an insulin injection device. It features a dose window, dose pointer, dose selector, rubber seal, insulin scale, insulin name, and injection button. The text notes that the plunger is not visible until after a few doses have been injected.*
Figure - toujeo 27

This appears to be a list of body parts - upper arms, stomach, and thighs. It is not clear what context this list is related to.*
Figure - toujeo 28

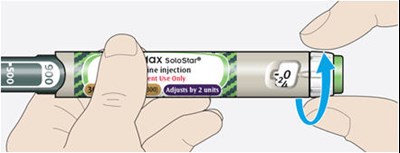
This text describes the components of a typical insulin pen used for injecting insulin. It mentions the cartridge holder, the dose window, dose pointer, dose selector, injection button, plunger, insulinscale, insulin name, pen cap and rubber seal. It also notes that the plunger will not be visible until a few doses are administered.*
PRINCIPAL DISPLAY PANEL - 1.5 mL Pen Carton - toujeo 51

This is the description for an insulin medication called Toujeo Solostar. It is indicated for subcutaneous injection only and should be used within 56 days of opening it. It comes in a disposable insulin delivery device and should not be removed with a syringe. It is not meant to be mixed with other insulins and should only be used if the solution is clear and colorless with no visible particles. This package includes three prefilled pens of 450 units each. The instructions warn to only use new needles and to store the unopened and opened device properly.*
PRINCIPAL DISPLAY PANEL - 3 mL Pen Carton - toujeo 52

This is a description of a insulin drug called "Toujeo Max Solostar". It is for single patient use only and is intended for subcutaneous injection only. It comes in a disposable insulin delivery device and should not be removed with a syringe. It is recommended to use a new needle every time. Mixing with other insulins is not allowed. It should only be used if the solution is clear and colorless with no particles visible, along with a security sticker. After opening, the solution should be used within 56 days. The text also contains instructions for storage and dosage, as well as information about needles that should be used.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.