Product Images Praluent
View Photos of Packaging, Labels & Appearance
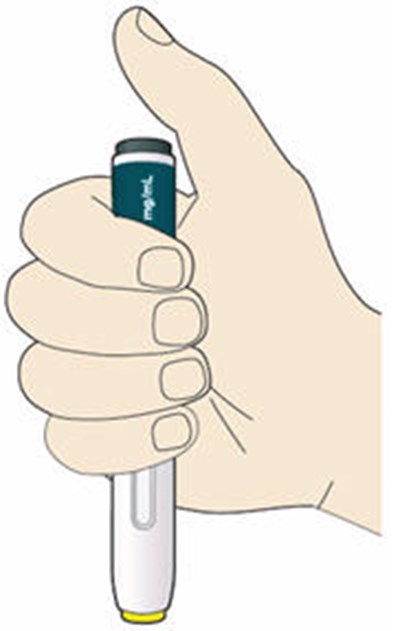
- Figure 1 - praluent 01
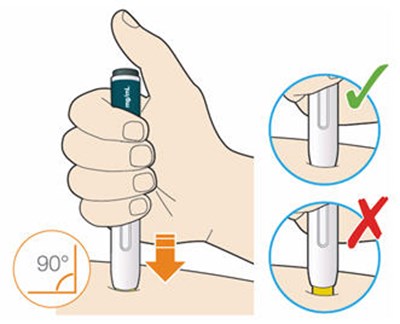
- Figure 2 - praluent 02
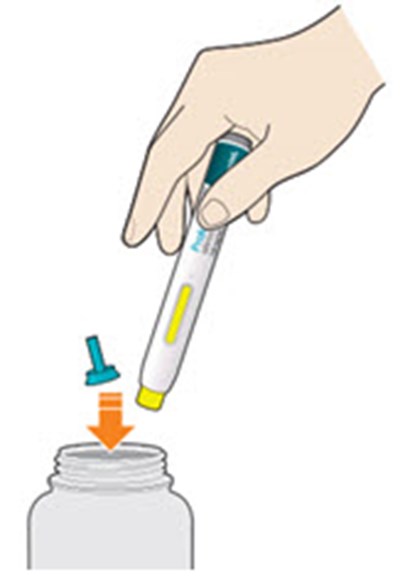
- Figure 3 - praluent 03
- Figure 4 - praluent 03a
- Figure 5 - praluent 03b
- Figure - praluent 04
- Figure - praluent 05
- Figure - praluent 06
- Figure - praluent 07
- Figure - praluent 08
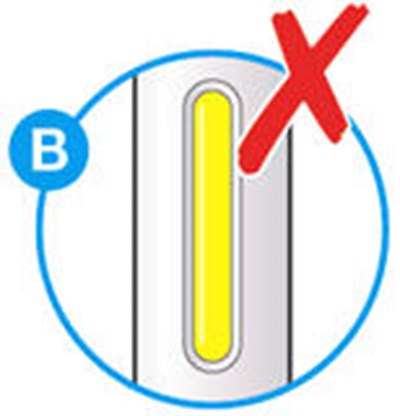
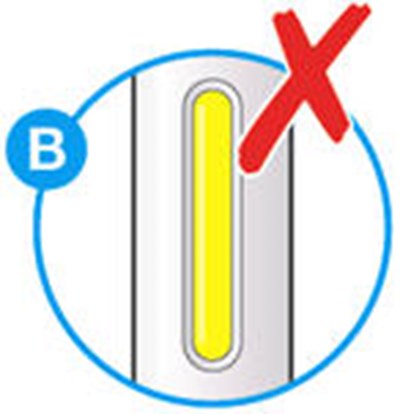
- Picture A - praluent 09
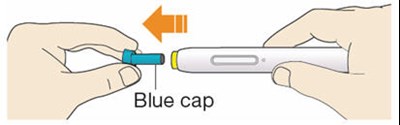
- Figure B - praluent 10
- Figure - praluent 11
- Figure - praluent 12
- Figure - praluent 13
- Figure - praluent 14
- Figure - praluent 15
- Figure - praluent 16
- Figure - praluent 17
- Figure - praluent 18
- Figure - praluent 19
- Figure - praluent 20
- Figure - praluent 21
- Figure - praluent 22
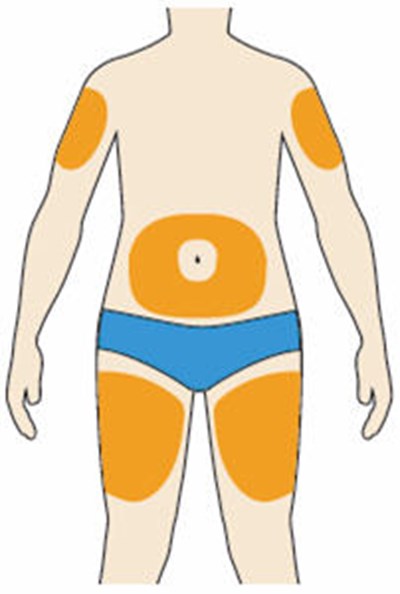
- Picture A - praluent 23
- Figure B - praluent 24
- Figure - praluent 25
- Figure - praluent 26
- Figure - praluent 27
- Figure - praluent 28
- Figure - praluent 29
- Figure - praluent 30
- Figure - praluent 31
- PRINCIPAL DISPLAY PANEL - 75 mg/mL Pen Carton - praluent 32
- PRINCIPAL DISPLAY PANEL - 150 mg/mL Pen Carton - praluent 33
- PRINCIPAL DISPLAY PANEL - 75 mg/mL Syringe Carton - praluent 34
- PRINCIPAL DISPLAY PANEL - 150 mg/mL Syringe Carton - praluent 35
Product Label Images
The following 37 images provide visual information about the product associated with Praluent NDC 0024-5901 by Sanofi-aventis U.s. Llc, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
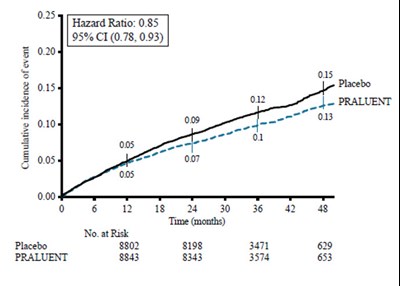
Figure 2 - praluent 02
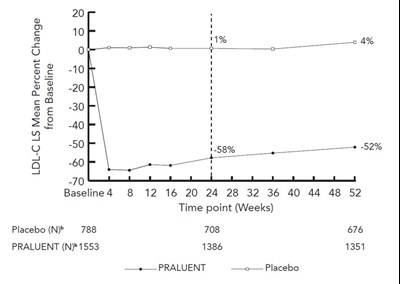
The text describes a table representing data related to the LS mean percent change from the baseline for LDL-C at different time points over 52 weeks for a treatment labeled PRALUENT (N) compared to a placebo treatment. The data shows a considerable decrease in LDL-C levels for the PRALUENT (N) treatment compared to the placebo treatment over the course of 52 weeks.*
Figure 3 - praluent 03

This appears to be a graph or chart showing the mean percent change of LDL-C before and after a treatment using a drug called PRALUENT compared to placebo. The graph shows that PRALUENT resulted in a decrease in LDL-C levels compared to placebo at various time points over a period of 52 weeks.*
Figure 4 - praluent 03a

This text appears to be a graph or chart that shows the LDL-C LS mean percent change from baseline for three groups over a period of 12 weeks, represented by time points on the x-axis. The groups are labeled as "Placebo," "Praluent75 Q2W," and "Praluent 300 Q4W," and have 157, 78, and 312 subjects, respectively. Without a visual, it is difficult to provide further details.*
Figure 5 - praluent 03b
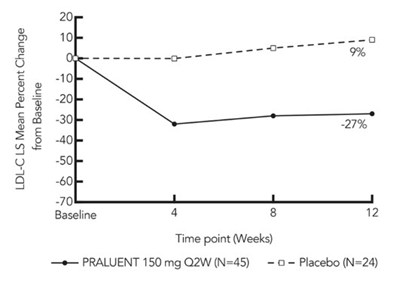
This is a table showing the LDL-C LS Mean Percent Change from Baseline over weeks 4, 8, and 12 for a study comparing PRALUENT 150 mg Q2W (N=45) to a placebo (N=24). The results indicate a -70% reduction in LDL-C for the PRALUENT group compared to the baseline.*
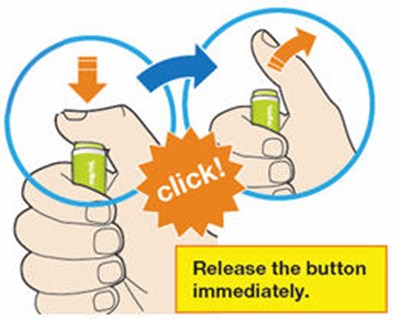
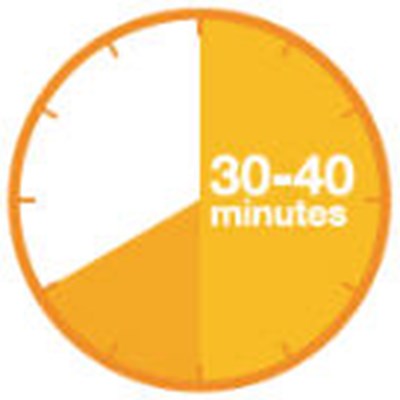
Figure - praluent 27
This is a simple instruction to be followed in a situation where a button has been pressed and needs to be released. The instruction is to release the button immediately.*
PRINCIPAL DISPLAY PANEL - 75 mg/mL Pen Carton - praluent 32

Not available. The given text contains mostly symbols, numerals, and some alphabets that are not intelligible.*
PRINCIPAL DISPLAY PANEL - 150 mg/mL Pen Carton - praluent 33

Not available. The text contains some words, but they do not make any sense and seem to be randomly arranged.*
PRINCIPAL DISPLAY PANEL - 75 mg/mL Syringe Carton - praluent 34

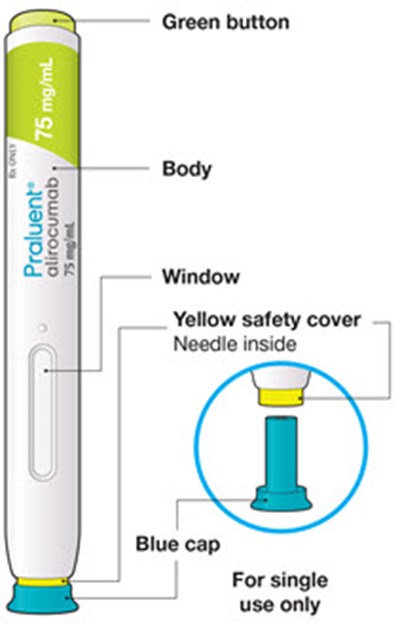
This is a description of Praluent, a medication used as an injection for subcutaneous administration only. It is available in a pre-filled syringe that contains 75mg/mL of the active ingredient alirocumab. The manufacturer of Praluent is Regeneron Sanofi. There is some blurry text at the end that is not readable.*
PRINCIPAL DISPLAY PANEL - 150 mg/mL Syringe Carton - praluent 35

The text is a product label of Praluent™ (alirocumab), an injection, containing 150mg/mL of the medication. It recommends the medication for subcutaneous infection only, meant for single-dose use. The label contains information about the manufacturer, dosage, date of infection, and usage instructions.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.