FDA Label for Dupixent
View Indications, Usage & Precautions
- 1 INDICATIONS AND USAGE
- 2.1 DOSAGE
- 2.2 IMPORTANT ADMINISTRATION INSTRUCTIONS
- 2.3 PREPARATION FOR USE OF DUPIXENT PRE-FILLED SYRINGE WITH NEEDLE SHIELD
- 3 DOSAGE FORMS AND STRENGTHS
- 4 CONTRAINDICATIONS
- 5.1 HYPERSENSITIVITY
- 5.2 CONJUNCTIVITIS AND KERATITIS
- 5.3 COMORBID ASTHMA
- 5.4 PARASITIC (HELMINTH) INFECTIONS
- 6 ADVERSE REACTIONS
- 6.1 CLINICAL TRIALS EXPERIENCE
- OTHER
- 6.2 IMMUNOGENICITY
- 7.1 LIVE VACCINES
- 7.2 NON-LIVE VACCINES
- 8.4 PEDIATRIC USE
- 8.5 GERIATRIC USE
- 10 OVERDOSE
- 11 DESCRIPTION
- 12.1 MECHANISM OF ACTION
- 12.2 PHARMACODYNAMICS
- 13.1 CARCINOGENESIS, MUTAGENESIS, IMPAIRMENT OF FERTILITY
- 14 CLINICAL STUDIES
- 16.1 HOW SUPPLIED
- 16.2 STORAGE AND HANDLING
- 17 PATIENT COUNSELING INFORMATION
- SPL PATIENT PACKAGE INSERT
- INSTRUCTIONS FOR USE
- PRINCIPAL DISPLAY PANEL - 300 MG/2 ML SYRINGE CARTON - NDC 0024-5914-01
- PRINCIPAL DISPLAY PANEL - 300 MG/2 ML SYRINGE CARTON - NDC 0024-5916-01
Dupixent Product Label
The following document was submitted to the FDA by the labeler of this product Sanofi-aventis U.s. Llc. The document includes published materials associated whith this product with the essential scientific information about this product as well as other prescribing information. Product labels may durg indications and usage, generic names, contraindications, active ingredients, strength dosage, routes of administration, appearance, warnings, inactive ingredients, etc.
1 Indications And Usage
DUPIXENT is indicated for the treatment of adult patients with moderate-to-severe atopic dermatitis whose disease is not adequately controlled with topical prescription therapies or when those therapies are not advisable. DUPIXENT can be used with or without topical corticosteroids.
2.1 Dosage
DUPIXENT is administered by subcutaneous injection.
The recommended dose of DUPIXENT for adult patients is an initial dose of 600 mg (two 300 mg injections), followed by 300 mg given every other week.
DUPIXENT can be used with or without topical corticosteroids. Topical calcineurin inhibitors may be used, but should be reserved for problem areas only, such as the face, neck, intertriginous and genital areas.
If a dose is missed, instruct the patient to administer the injection within 7 days from the missed dose and then resume the patient's original schedule. If the missed dose is not administered within 7 days, instruct the patient to wait until the next dose on the original schedule.
2.2 Important Administration Instructions
DUPIXENT is intended for use under the guidance of a healthcare provider. A patient may self-inject DUPIXENT after training in subcutaneous injection technique using the pre-filled syringe. Provide proper training to patients and/or caregivers on the preparation and administration of DUPIXENT prior to use according to the "Instructions for Use".
For the initial 600 mg dose, administer each of the two DUPIXENT 300 mg injections at different injection sites.
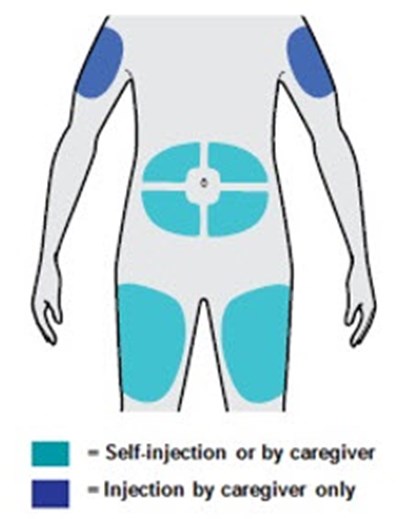
Administer subcutaneous injection into the thigh or abdomen, except for the 2 inches (5 cm) around the navel. The upper arm can also be used if a caregiver administers the injection.
Rotate the injection site with each injection. DO NOT inject DUPIXENT into skin that is tender, damaged, bruised, or scarred.
The DUPIXENT "Instructions for Use" contains more detailed instructions on the preparation and administration of DUPIXENT [see Instructions for Use].
2.3 Preparation For Use Of Dupixent Pre-Filled Syringe With Needle Shield
Before injection, remove DUPIXENT pre-filled syringe from the refrigerator and allow DUPIXENT to reach room temperature (45 minutes) without removing the needle cap.
Inspect DUPIXENT visually for particulate matter and discoloration prior to administration. DUPIXENT is a clear to slightly opalescent, colorless to pale yellow solution. Do not use if the liquid contains visible particulate matter, is discolored or cloudy (other than clear to slightly opalescent, colorless to pale yellow). DUPIXENT does not contain preservatives; therefore, discard any unused product remaining in the pre-filled syringe.
3 Dosage Forms And Strengths
DUPIXENT is a clear to slightly opalescent, colorless to pale yellow solution available as:
- Injection: 300 mg/2 mL in a single-dose pre-filled syringe with needle shield
4 Contraindications
DUPIXENT is contraindicated in patients who have known hypersensitivity to dupilumab or any of its excipients [see Warnings and Precautions (5.1)].
5.1 Hypersensitivity
Hypersensitivity reactions, including generalized urticaria and serum sickness or serum sickness-like reactions, were reported in less than 1% of subjects who received DUPIXENT in clinical trials. Two subjects experienced serum sickness or serum sickness-like reactions that were associated with high titers of antibodies to dupilumab [see Adverse Reactions (6.2)]. If a clinically significant hypersensitivity reaction occurs, institute appropriate therapy and discontinue DUPIXENT [see Adverse Reactions (6.1, 6.2)].
5.2 Conjunctivitis And Keratitis
Conjunctivitis and keratitis occurred more frequently in subjects who received DUPIXENT. Conjunctivitis was the most frequently reported eye disorder. Most subjects with conjunctivitis recovered or were recovering during the treatment period [see Adverse Reactions (6.1)].
Keratitis was reported in <1% of the DUPIXENT group (1 per 100 subject-years) and in 0% of the placebo group (0 per 100 subject-years) in the 16-week monotherapy trials. In the 52-week DUPIXENT + topical corticosteroids (TCS) trial, keratitis was reported in 4% of the DUPIXENT + TCS group (12 per 100 subject-years) and in 0% of the placebo + TCS group (0 per 100 subject-years). Most subjects with keratitis recovered or were recovering during the treatment period [see Adverse Reactions (6.1)].
Advise patients to report new onset or worsening eye symptoms to their healthcare provider.
5.3 Comorbid Asthma
Safety and efficacy of DUPIXENT have not been established in the treatment of asthma. Advise patients with comorbid asthma not to adjust or stop their asthma treatments without consultation with their physicians.
5.4 Parasitic (Helminth) Infections
Patients with known helminth infections were excluded from participation in clinical studies. It is unknown if DUPIXENT will influence the immune response against helminth infections.
6 Adverse Reactions
The following adverse reactions are discussed in greater detail elsewhere in the labeling:
- Hypersensitivity [see Warnings and Precautions (5.1)]
- Conjunctivitis and Keratitis [see Warnings and Precautions (5.2)]
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
Three randomized, double-blind, placebo-controlled, multicenter trials (Trials 1, 2, and 3) and one dose-ranging trial (Trial 4) evaluated the safety of DUPIXENT in subjects with moderate-to-severe atopic dermatitis. The safety population had a mean age of 38 years; 41% of subjects were female, 67% were white, 24% were Asian, and 6% were black; in terms of comorbid conditions, 48% of the subjects had asthma, 49% had allergic rhinitis, 37% had food allergy, and 27% had allergic conjunctivitis. In these 4 trials, 1472 subjects were treated with subcutaneous injections of DUPIXENT, with or without concomitant topical corticosteroids (TCS).
A total of 739 subjects were treated with DUPIXENT for at least 1 year in the development program for moderate-to-severe atopic dermatitis.
Trials 1, 2, and 4 compared the safety of DUPIXENT monotherapy to placebo through Week 16. Trial 3 compared the safety of DUPIXENT + TCS to placebo + TCS through Week 52.
Other
Weeks 0 to 16 (Trials 1 to 4):
In DUPIXENT monotherapy trials (Trials 1, 2, and 4) through Week 16, the proportion of subjects who discontinued treatment because of adverse events was 1.9% in both the DUPIXENT 300 mg Q2W and placebo groups.
Table 1 summarizes the adverse reactions that occurred at a rate of at least 1% in the DUPIXENT 300 mg Q2W monotherapy groups, and in the DUPIXENT + TCS group, all at a higher rate than in their respective comparator groups during the first 16 weeks of treatment.
| DUPIXENT Monotherapy pooled analysis of Trials 1, 2, and 4 | DUPIXENT + TCS analysis of Trial 3 where subjects were on background TCS therapy | |||
|---|---|---|---|---|
| Adverse Reaction | DUPIXENT 300 mg Q2W DUPIXENT 600 mg at Week 0, followed by 300 mg every two weeks | Placebo | DUPIXENT 300 mg Q2W | Placebo + TCS |
| N=529 n (%) | N=517 n (%) | N=110 n (%) | N=315 n (%) | |
| Injection site reactions | 51 (10) | 28 (5) | 11 (10) | 18 (6) |
| Conjunctivitis Conjunctivitis cluster includes conjunctivitis, allergic conjunctivitis, bacterial conjunctivitis, viral conjunctivitis, giant papillary conjunctivitis, eye irritation, and eye inflammation. | 51 (10) | 12 (2) | 10 (9) | 15 (5) |
| Blepharitis | 2 (<1) | 1 (<1) | 5 (5) | 2 (1) |
| Oral herpes | 20 (4) | 8 (2) | 3 (3) | 5 (2) |
| Keratitis Keratitis cluster includes keratitis, ulcerative keratitis, allergic keratitis, atopic keratoconjunctivitis, and ophthalmic herpes simplex. | 1 (<1) | 0 | 4 (4) | 0 |
| Eye pruritus | 3 (1) | 1 (<1) | 2 (2) | 2 (1) |
| Other herpes simplex virus infection Other herpes simplex virus infection cluster includes herpes simplex, genital herpes, herpes simplex otitis externa, and herpes virus infection, but excludes eczema herpeticum. | 10 (2) | 6 (1) | 1 (1) | 1 (<1) |
| Dry eye | 1 (<1) | 0 | 2 (2) | 1 (<1) |
Safety through Week 52 (Trial 3):
In the DUPIXENT with concomitant TCS trial (Trial 3) through Week 52, the proportion of subjects who discontinued treatment because of adverse events was 1.8% in DUPIXENT 300 mg Q2W + TCS group and 7.6% in the placebo + TCS group. Two subjects discontinued DUPIXENT because of adverse reactions: atopic dermatitis (1 subject) and exfoliative dermatitis (1 subject).
The safety profile of DUPIXENT + TCS through Week 52 was generally consistent with the safety profile observed at Week 16.
Specific Adverse Reactions
Conjunctivitis
During the 52-week treatment period of concomitant therapy trial (Trial 3), conjunctivitis was reported in 16% of the DUPIXENT 300 mg Q2W + TCS group (20 per 100 subject-years) and in 9% of the placebo + TCS group (10 per 100 subject-years) [see Warnings and Precautions (5.2)].
Eczema Herpeticum and Herpes Zoster
The rate of eczema herpeticum was similar in the placebo and DUPIXENT groups.
Herpes zoster was reported in <0.1% of the DUPIXENT groups (<1 per 100 subject-years) and in <1% of the placebo group (1 per 100 subject-years) in the 16-week monotherapy trials. In the 52-week DUPIXENT + TCS trial, herpes zoster was reported in 1% of the DUPIXENT + TCS group (1 per 100 subject-years) and 2% of the placebo + TCS group (2 per 100 subject-years).
Hypersensitivity Reactions
Hypersensitivity reactions were reported in <1% of DUPIXENT-treated subjects. These included serum sickness reaction, serum sickness-like reaction, and generalized urticaria [see Contraindications (4), Warnings and Precautions (5.1), and Adverse Reactions (6.2)].
Eosinophils
DUPIXENT-treated subjects had a greater mean initial increase from baseline in eosinophil count compared to subjects treated with placebo in the monotherapy trials. Eosinophil counts declined to near baseline levels by Week 16. The initial increase in eosinophils was not observed in the 52-week DUPIXENT + TCS trial.
In Trials 1, 2, and 3, the incidence of treatment-emergent eosinophilia (≥500 cells/mcL) was similar in DUPIXENT and placebo groups. In Trials 1, 2, and 3, treatment-emergent eosinophilia (≥5,000 cells/mcL) was reported in <1% of DUPIXENT-treated patients and none in placebo-treated patients. In most cases, eosinophil counts declined to near baseline during study treatment.
Risk Summary
There are no available data on DUPIXENT use in pregnant women to inform any drug associated risk. Human IgG antibodies are known to cross the placental barrier; therefore, DUPIXENT may be transmitted from the mother to the developing fetus. In an enhanced pre- and post-natal developmental study, no adverse developmental effects were observed in offspring born to pregnant monkeys after subcutaneous administration of a homologous antibody against interleukin-4-receptor alpha (IL-4Rα) during organogenesis through parturition at doses up to 10-times the maximum recommended human dose (MRHD) [see Data]. The estimated background risk of major birth defects and miscarriage for the indicated population is unknown. All pregnancies have a background risk of birth defect, loss or other adverse outcomes. In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2% to 4% and 15% to 20%, respectively.
Data
Animal Data
In an enhanced pre- and post-natal development toxicity study, pregnant cynomolgus monkeys were administered weekly subcutaneous doses of homologous antibody against IL-4Rα up to 10 times the MRHD (on a mg/kg basis of 100 mg/kg/week) from the beginning of organogenesis to parturition. No treatment-related adverse effects on embryofetal toxicity or malformations, or on morphological, functional, or immunological development were observed in the infants from birth through 6 months of age.
Risk Summary
There are no data on the presence of dupilumab in human milk, the effects on the breastfed infant, or the effects on milk production. Human IgG is known to be present in human milk. The effects of local gastrointestinal and limited systemic exposure to dupilumab on the breastfed infant are unknown. The developmental and health benefits of breastfeeding should be considered along with the mother's clinical need for DUPIXENT and any potential adverse effects on the breastfed child from DUPIXENT or from the underlying maternal condition.
Absorption
Following an initial subcutaneous (SC) dose of 600 mg, dupilumab reached peak mean ±SD concentrations (Cmax) of 70.1±24.1 mcg/mL by approximately 1 week post dose.
Steady-state concentrations were achieved by Week 16 following the administration of 600 mg starting dose and 300 mg dose either weekly (twice the recommended dosing frequency) or every other week. Across clinical trials, the mean ±SD steady-state trough concentrations ranged from 73.3±40.0 mcg/mL to 79.9±41.4 mcg/mL for 300 mg administered every 2 weeks and from 173±75.9 mcg/mL to 193±77.0 mcg/mL for 300 mg administered weekly.
The bioavailability of dupilumab following a SC dose is estimated to be 64%.
Distribution
The estimated total volume of distribution was approximately 4.8±1.3 L.
Elimination
The metabolic pathway of dupilumab has not been characterized. As a human monoclonal IgG4 antibody, dupilumab is expected to be degraded into small peptides and amino acids via catabolic pathways in the same manner as endogenous IgG. After the last steady-state dose of 300 mg Q2W or 300 mg QW dupilumab, the median times to non-detectable concentration (<78 ng/mL) are 10 and 13 weeks, respectively.
Dose Linearity
Dupilumab exhibited nonlinear target-mediated pharmacokinetics with exposures increasing in a greater than dose-proportional manner. The systemic exposure increased by 30-fold when the dose increased 8-fold following a single dose of dupilumab from 75 mg to 600 mg (i.e., 0.25-times to 2-times the recommended dose).
Weight
Dupilumab trough concentrations were lower in subjects with higher body weight.
Immunogenicity
Development of antibodies to dupilumab was associated with lower serum dupilumab concentrations. A few subjects who had high antibody titers also had no detectable serum dupilumab concentrations.
Specific Populations
Geriatric Patients
In subjects who are 65 years and older, the mean ±SD steady-state trough concentrations of dupilumab were 69.4±31.4 mcg/mL and 166±62.3 mcg/mL, respectively, for 300 mg administered every 2 weeks and weekly. No dose adjustment in this population is recommended.
Renal or Hepatic Impairment
No formal trial of the effect of hepatic or renal impairment on the pharmacokinetics of dupilumab was conducted.
Drug Interaction Studies
Cytochrome P450 Substrates
The effects of dupilumab on the pharmacokinetics of midazolam (metabolized by CYP3A4), warfarin (metabolized by CYP2C9), omeprazole (metabolized by CYP2C19), metoprolol (metabolized by CYP2D6), and caffeine (metabolized by CYP1A2) were evaluated in a study with 12-13 evaluable subjects with atopic dermatitis (a SC loading dose of 600 mg followed by 300 mg SC weekly for six weeks). No clinically significant changes in AUC were observed. The largest effect was observed for metoprolol (CYP2D6) with an increase in AUC of 29%.
Clinical Response at Week 16 (Trials 1, 2, and 3)
The results of the DUPIXENT monotherapy trials (Trials 1 and 2) and the DUPIXENT with concomitant TCS trial (Trial 3) are presented in Table 2.
| Trial 1 | Trial 2 | Trial 3 | ||||
|---|---|---|---|---|---|---|
| DUPIXENT 300 mg Q2W | Placebo | DUPIXENT 300 mg Q2W | Placebo | DUPIXENT 300 mg Q2W + TCS | Placebo + TCS | |
| Number of subjects randomized (FAS) Full Analysis Set (FAS) includes all subjects randomized. | 224 | 224 | 233 | 236 | 106 | 315 |
| IGA 0 or 1 Responder was defined as a subject with IGA 0 or 1 ("clear" or "almost clear") with a reduction of ≥2 points on a 0-4 IGA scale. ,Subjects who received rescue treatment or with missing data were considered as non-responders. | 38% | 10% | 36% | 9% | 39% | 12% |
| EASI-75 | 51% | 15% | 44% | 12% | 69% | 23% |
| EASI-90 | 36% | 8% | 30% | 7% | 40% | 11% |
| Number of subjects with baseline Peak Pruritus NRS score ≥4 | 213 | 212 | 225 | 221 | 102 | 299 |
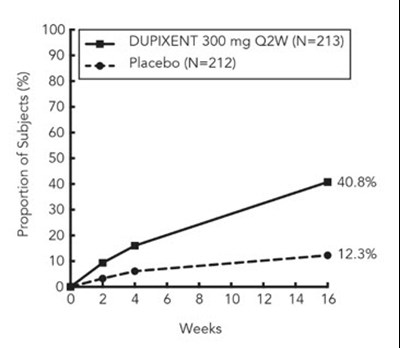
| Peak Pruritus NRS (≥4-point improvement) | 41% | 12% | 36% | 10% | 59% | 20% |
| Trial 1 | Trial 2 |
|---|---|
| a In the primary analyses of the efficacy endpoints, subjects who received rescue treatment or with missing data were considered non-responders. | |
| b Full Analysis Set (FAS) includes all subjects randomized. | |
In Trial 3, of the 421 subjects, 353 had been on study for 52 weeks at the time of data analysis. Of these 353 subjects, responders at Week 52 represent a mixture of subjects who maintained their efficacy from Week 16 (e.g., 53% of DUPIXENT IGA 0 or 1 responders at Week 16 remained responders at Week 52) and subjects who were non-responders at Week 16 who later responded to treatment (e.g., 24% of DUPIXENT IGA 0 or 1 non-responders at Week 16 became responders at Week 52). Results of supportive analyses of the 353 subjects in the DUPIXENT with concomitant TCS trial (Trial 3) are presented in Table 3.
| DUPIXENT 300 mg Q2W + TCS | Placebo + TCS | |
|---|---|---|
| Number of Subjects In Trial 3, of the 421 randomized and treated subjects, 68 subjects (16%) had not been on study for 52 weeks at the time of data analysis. | 89 | 264 |
| Responder Responder was defined as a subject with IGA 0 or 1 ("clear" or "almost clear") with a reduction of ≥2 points on a 0-4 IGA scale. ,Subjects who received rescue treatment or with missing data were considered as non-responders. at Week 16 and 52 | 22% | 7% |
| Responder at Week 16 but Non-responder at Week 52 | 20% | 7% |
| Non-responder at Week 16 and Responder at Week 52 | 13% | 6% |
| Non-responder at Week 16 and 52 | 44% | 80% |
| Overall Responder | 36% | 13% |
Treatment effects in subgroups (weight, age, gender, race, and prior treatment, including immunosuppressants) in Trials 1, 2, and 3 were generally consistent with the results in the overall study population.
In Trials 1, 2, and 3, a third randomized treatment arm of DUPIXENT 300 mg QW did not demonstrate additional treatment benefit over DUPIXENT 300 mg Q2W.
Subjects in Trials 1 and 2 who had an IGA 0 or 1 with a reduction of ≥2 points were re-randomized into Trial 5. Trial 5 evaluated multiple DUPIXENT monotherapy dose regimens for maintaining treatment response. The study included subjects randomized to continue with DUPIXENT 300 mg Q2W (62 subjects) or switch to placebo (31 subjects) for 36 weeks. IGA 0 or 1 responses at Week 36 were as follows: 33 (53%) in the Q2W group and 3 (10%) in the placebo group.
Administration Instructions
Provide proper training to patients and/or caregivers on proper subcutaneous injection technique, including aseptic technique, and the preparation and administration of DUPIXENT prior to use. Advise patients to follow sharps disposal recommendations [see Instructions for Use].
Hypersensitivity
Advise patients to discontinue DUPIXENT and to seek immediate medical attention if they experience any symptoms of systemic hypersensitivity reactions [see Warnings and Precautions (5.1)].
Conjunctivitis and Keratitis
Advise patients to consult their healthcare provider if new onset or worsening eye symptoms develop [see Warnings and Precautions (5.2)].
Comorbid Asthma
Advise patients with comorbid asthma not to adjust or stop their asthma treatment without talking to their physicians [see Warnings and Precautions (5.3)].
REGENERON SANOFI GENZYME
Manufactured by:
Regeneron Pharmaceuticals, Inc.
Tarrytown, NY 10591
U.S. License No. 1760
Marketed by:
sanofi-aventis U.S. LLC (Bridgewater, NJ 08807) and
Regeneron Pharmaceuticals, Inc. (Tarrytown, NY 10591)
DUPIXENT® is a registered trademark of Sanofi Biotechnology
© 2017 Regeneron Pharmaceuticals, Inc. / sanofi-aventis U.S. LLC. All rights reserved.
Revised: April 2018
Initial U.S. Approval: 2017
6.2 Immunogenicity
As with all therapeutic proteins, there is a potential for immunogenicity. The detection of antibody formation is highly dependent on the sensitivity and specificity of the assay. Additionally, the observed incidence of antibody (including neutralizing antibody) positivity in an assay may be influenced by several factors, including assay methodology, sample handling, timing of sample collection, concomitant medications, and underlying disease. For these reasons, comparison of the incidence of antibodies to dupilumab in the studies described below with the incidence of antibodies in other studies or to other products may be misleading.
Approximately 7% of subjects with atopic dermatitis who received DUPIXENT 300 mg Q2W for 16 weeks developed antibodies to dupilumab. Of the subjects who developed antibodies to dupilumab, approximately 30% (2% of all subjects receiving DUPIXENT) had antibodies that were classified as neutralizing.
Of the subjects with atopic dermatitis who received DUPIXENT 300 mg Q2W + TCS for 52 weeks, approximately 7% developed antibodies to dupilumab and approximately 2% had persistent antibody responses, defined as having at least 2 consecutive positive post-baseline samples. Of the subjects who developed antibodies to dupilumab, approximately 14% (1% of all subjects receiving DUPIXENT + TCS) had antibodies that were classified as neutralizing.
In subjects who received DUPIXENT, development of antibodies to dupilumab was associated with lower serum dupilumab concentrations [see Clinical Pharmacology (12.3)].
Antibodies to dupilumab were detected in approximately 2% and 8% of subjects with atopic dermatitis in the placebo or the placebo + TCS groups, respectively.
The antibody titers detected in both DUPIXENT and placebo subjects were generally low. Two subjects developed serum sickness or serum sickness-like reactions and high titers of antibodies to dupilumab during DUPIXENT therapy [see Warnings and Precautions (5.1)].
7.1 Live Vaccines
Avoid use of live vaccines in patients treated with DUPIXENT.
7.2 Non-Live Vaccines
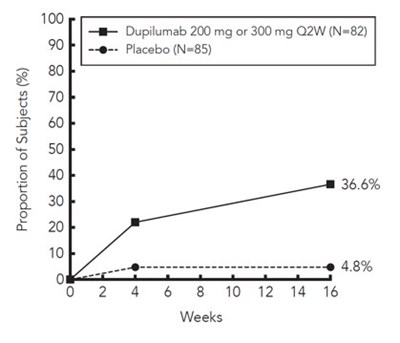
Immune responses to vaccination were assessed in a study in which subjects with atopic dermatitis were treated once weekly for 16 weeks with 300 mg of dupilumab (twice the recommended dosing frequency). After 12 weeks of DUPIXENT administration, subjects were vaccinated with a Tdap vaccine (Adacel®) and a meningococcal polysaccharide vaccine (Menomune®). Antibody responses to tetanus toxoid and serogroup C meningococcal polysaccharide were assessed 4 weeks later. Antibody responses to both tetanus vaccine and meningococcal polysaccharide vaccine were similar in dupilumab-treated and placebo-treated subjects. Immune responses to the other active components of the Adacel and Menomune vaccines were not assessed.
8.4 Pediatric Use
Safety and efficacy in pediatric patients (<18 years of age) have not been established.
8.5 Geriatric Use
Of the 1472 subjects with atopic dermatitis exposed to DUPIXENT in a dose-ranging study and placebo-controlled trials, 67 subjects were 65 years or older. Although no differences in safety or efficacy were observed between older and younger subjects, the number of subjects aged 65 and over is not sufficient to determine whether they respond differently from younger subjects [see Clinical Pharmacology (12.3)].
10 Overdose
There is no specific treatment for DUPIXENT overdose. In the event of overdosage, monitor the patient for any signs or symptoms of adverse reactions and institute appropriate symptomatic treatment immediately.
11 Description
Dupilumab, an interleukin-4 receptor alpha antagonist, is a human monoclonal antibody of the IgG4 subclass that binds to the IL-4Rα subunit and inhibits IL-4 and IL-13 signaling. Dupilumab has an approximate molecular weight of 147 kDa.
Dupilumab is produced by recombinant DNA technology in Chinese Hamster Ovary cell suspension culture.
DUPIXENT (dupilumab) Injection is supplied as a sterile, preservative-free, clear to slightly opalescent, colorless to pale yellow solution for subcutaneous injection. DUPIXENT is provided as a single-dose pre-filled syringe with needle shield in a 2.25 mL siliconized Type-1 clear glass syringe. The needle cap is not made with natural rubber latex. Each pre-filled syringe delivers 300 mg dupilumab in 2 mL which also contains L-arginine hydrochloride (10.5 mg), L-histidine (6.2 mg), polysorbate 80 (4 mg), sodium acetate (2 mg), sucrose (100 mg), and water for injection, pH 5.9.
12.1 Mechanism Of Action
Dupilumab is a human monoclonal IgG4 antibody that inhibits interleukin-4 (IL-4) and interleukin-13 (IL-13) signaling by specifically binding to the IL-4Rα subunit shared by the IL-4 and IL-13 receptor complexes. Dupilumab inhibits IL-4 signaling via the Type I receptor and both IL-4 and IL-13 signaling through the Type II receptor.
Blocking IL-4Rα with dupilumab inhibits IL-4 and IL-13 cytokine-induced responses, including the release of proinflammatory cytokines, chemokines and IgE.
12.2 Pharmacodynamics
Consistent with receptor blockade, serum levels of IL-4 and IL-13 were increased following dupilumab treatment. The relationship between the pharmacodynamic activity and the mechanism(s) by which dupilumab exerts its clinical effects is unknown.
13.1 Carcinogenesis, Mutagenesis, Impairment Of Fertility
Animal studies have not been conducted to evaluate the carcinogenic or mutagenic potential of dupilumab.
No effects on fertility parameters such as reproductive organs, menstrual cycle length, or sperm analysis were observed in sexually mature mice that were subcutaneously administered a homologous antibody against IL-4Rα at doses up to 200 mg/kg/week.
14 Clinical Studies
Three randomized, double-blind, placebo-controlled trials (Trials 1, 2, and 3) enrolled a total of 2119 subjects 18 years of age and older with moderate-to-severe atopic dermatitis (AD) not adequately controlled by topical medication(s). Disease severity was defined by an Investigator's Global Assessment (IGA) score ≥3 in the overall assessment of AD lesions on a severity scale of 0 to 4, an Eczema Area and Severity Index (EASI) score ≥16 on a scale of 0 to 72, and a minimum body surface area involvement of ≥10%. At baseline, 59% of subjects were male, 67% were white, 52% of subjects had a baseline IGA score of 3 (moderate AD), and 48% of subjects had a baseline IGA of 4 (severe AD). The baseline mean EASI score was 33 and the baseline weekly averaged peak pruritus Numeric Rating Scale (NRS) was 7 on a scale of 0-10.
In all three trials, subjects in the DUPIXENT group received subcutaneous injections of DUPIXENT 600 mg at Week 0, followed by 300 mg every other week (Q2W). In the monotherapy trials (Trials 1 and 2), subjects received DUPIXENT or placebo for 16 weeks.
In the concomitant therapy trial (Trial 3), subjects received DUPIXENT or placebo with concomitant topical corticosteroids (TCS) and as needed topical calcineurin inhibitors for problem areas only, such as the face, neck, intertriginous and genital areas for 52 weeks.
All three trials assessed the primary endpoint, the change from baseline to Week 16 in the proportion of subjects with an IGA 0 (clear) or 1 (almost clear) and at least a 2-point improvement. Other endpoints included the proportion of subjects with EASI-75 (improvement of at least 75% in EASI score from baseline), and reduction in itch as defined by at least a 4-point improvement in the peak pruritus NRS from baseline to Week 16.
16.1 How Supplied
DUPIXENT (dupilumab) Injection is a clear to slightly opalescent, colorless to pale yellow solution, supplied in single-dose pre-filled syringes with needle shield. Each pre-filled syringe with needle shield is designed to deliver 300 mg of DUPIXENT in 2 mL solution.
DUPIXENT is available in cartons containing 2 pre-filled syringes with needle shield.
| Pack Size | 300 mg/2 mL Pre-filled Syringe with Needle Shield |
| Pack of 2 syringes | NDC 0024-5914-01 |
16.2 Storage And Handling
DUPIXENT is sterile and preservative-free. Discard any unused portion.
Store refrigerated at 36°F to 46°F (2°C to 8°C) in the original carton to protect from light.
If necessary, pre-filled syringes may be kept at room temperature up to 77°F (25°C) for a maximum of 14 days. Do not store above 77°F (25°C). After removal from the refrigerator, DUPIXENT must be used within 14 days or discarded.
Do not expose the syringe to heat or direct sunlight.
Any unused medicinal product or waste material should be disposed of in accordance with local requirements.
Do NOT freeze. Do NOT expose to heat. Do NOT shake.
17 Patient Counseling Information
Advise the patients and/or caregivers to read the FDA-approved patient labeling (Patient Information and Instructions for Use) before the patient starts using DUPIXENT and each time the prescription is renewed as there may be new information they need to know.
Spl Patient Package Insert
| Patient Information DUPIXENT® (DU-pix'-ent) (dupilumab) Injection, for subcutaneous use | |||
|---|---|---|---|
| This Patient Information has been approved by the U.S. Food and Drug Administration. | Issued: March 2017 | ||
What is DUPIXENT?
| |||
Do not use DUPIXENT if you are allergic to dupilumab or to any of the ingredients in DUPIXENT. See the end of this leaflet for a complete list of ingredients in DUPIXENT. | |||
Before using DUPIXENT, tell your healthcare provider about all your medical conditions, including if you:
Tell your healthcare provider about all of the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements. If you have asthma and are taking asthma medicines, do not change or stop your asthma medicine without talking to your healthcare provider. | |||
How should I use DUPIXENT?
| |||
What are the possible side effects of DUPIXENT? DUPIXENT can cause serious side effects, including:
| |||
|
|
| |
The most common side effects of DUPIXENT include: | |||
|
| ||
Tell your healthcare provider if you have any side effect that bothers you or that does not go away. These are not all of the possible side effects of DUPIXENT. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. | |||
General information about the safe and effective use of DUPIXENT. Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not use DUPIXENT for a condition for which it was not prescribed. Do not give DUPIXENT to other people, even if they have the same symptoms that you have. It may harm them. You can ask your pharmacist or healthcare provider for information about DUPIXENT that is written for health professionals. | |||
What are the ingredients in DUPIXENT? Active ingredient: dupilumab Inactive ingredients: L-arginine hydrochloride, L-histidine, polysorbate 80, sodium acetate, sucrose, and water for injection. | |||
REGENERON SANOFI GENZYME Manufactured by: Regeneron Pharmaceuticals, Inc., Tarrytown, NY 10591 U.S. License No. 1760 Marketed by: sanofi-aventis U.S. LLC (Bridgewater, NJ 08807) and Regeneron Pharmaceuticals, Inc. (Tarrytown, NY 10591) DUPIXENT® is a registered trademark of Sanofi Biotechnology / © 2017 Regeneron Pharmaceuticals, Inc. / sanofi-aventis U.S. LLC. All rights reserved. For more information about DUPIXENT, go to www.DUPIXENT.com or call 1- 844-DUPIXENT (1-844-387-4936). | |||
Instructions For Use
DUPIXENT® (DU-pix'-ent)
(dupilumab)
Injection, for subcutaneous use
Single-Dose Pre-filled Syringe with Needle Shield
Read this Instructions for Use before using the DUPIXENT Pre-filled Syringe. Do not inject yourself or someone else until you have been shown how to inject DUPIXENT. Your healthcare provider can show you or your caregiver how to prepare and inject a dose of DUPIXENT before you try to do it yourself the first time. Keep these instructions for future use. Call your healthcare provider if you have any questions.
This device is a Single-Dose Pre-filled Syringe (called "DUPIXENT Syringe" in these instructions). It contains 300 mg of DUPIXENT for injection under the skin (subcutaneous injection).
The parts of the DUPIXENT Syringe are shown below:
| Important Information | |
|---|---|
|
|
How should I store DUPIXENT?
| |
Step 1: Remove
Remove the DUPIXENT Syringe from the carton by holding the middle of the Syringe Body.
Do not pull off the Needle Cap until you are ready to inject.
Do not use the DUPIXENT Syringe if it has been dropped on a hard surface or damaged.
Step 2: Prepare
Ensure you have the following:
|
Step 3: Inspect
When you receive your DUPIXENT Syringes, always check to see that:
- you have the correct medicine and dose.
- the expiration date on the Single-Dose Pre-filled Syringe has not passed.
- You can inject into your thigh or stomach, except for the 2 inches (5 cm) around your belly button (navel).
- If a caregiver injects your dose, they can also use the outer area of the upper arm.
- Choose a different site each time you inject DUPIXENT.
- made of a heavy-duty plastic,
- can be closed with a tight-fitting, puncture-resistant lid, without sharps being able to come out,
- upright and stable during use,
- leak-resistant, and
- properly labeled to warn of hazardous waste inside the container
Do not use the DUPIXENT Syringe if the expiration date has passed.
Look at the medicine through the Viewing Window on the DUPIXENT Syringe:
Check to see if the liquid is clear and colorless to pale yellow.
Note: You may see an air bubble, this is normal.
Do not use the DUPIXENT Syringe if the liquid is discolored or cloudy, or if it contains visible flakes or particles.
Step 4: Wait 45 minutes
Lay the DUPIXENT Syringe on a flat surface and let it naturally warm to room temperature for at least 45 minutes.
Do not heat the DUPIXENT Syringe.
Do not put the DUPIXENT Syringe into direct sunlight.
Do not keep DUPIXENT Syringes at room temperature for more than 14 days. Throw away (dispose of) any DUPIXENT Syringes that have been left at room temperature for longer than 14 days.
Step 5: Choose your injection site
Do not inject into skin that is tender, damaged, bruised or scarred.
Step 6: Clean
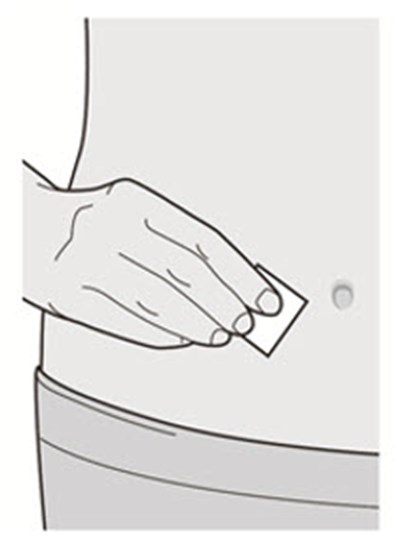
Wash your hands.
Clean the injection site with an alcohol wipe.
Let your skin dry before injecting.
Do not touch the injection site again or blow on it before the injection.
Step 7: Remove Needle Cap
Hold the DUPIXENT Syringe in the middle of the Syringe Body with the Needle pointing away from you and pull off the Needle Cap.
Do not put the Needle Cap back on.
Do not touch the Needle.
Inject your medicine right away after removing the Needle Cap.
Step 8: Pinch
Pinch a fold of skin at the injection site (thigh or stomach, except 2 inches around your belly button, or outer area of the upper arm if injected by your caregiver). The figure below shows an example of pinching a fold of skin on your stomach.
Step 9: Insert
Insert the Needle completely into the fold of the skin at about a 45° angle.
Step 10: Push
Relax the pinch.
Push the Plunger Rod down slowly and steadily as far as it will go until the DUPIXENT Syringe is empty.
Note: You will feel some resistance. This is normal.
Step 11: Remove
Keep pressing down on the Plunger and remove the Needle from the skin at the same angle it was inserted.
Do not put the Needle Cap back on.
Step 12: Release
Once the Needle is out of the skin, lift your thumb to retract the Needle up into the Needle Shield.
Lightly press a cotton ball or gauze on the injection site if you see any blood.
Do not rub your skin after the injection.
Step 13: Dispose
Put your used Needles, DUPIXENT Syringes, and Needle Caps in a FDA-cleared sharps disposal container right away after use.
Do not dispose of (throw away) Needles, DUPIXENT Syringes, and Needle Caps in your household trash.
If you do not have a FDA-cleared sharps disposal container, you may use a household container that is:
When your sharps disposal container is almost full, you will need to follow your community guidelines for the right way to dispose of your sharps disposal container. There may be state or local laws about how you should throw away used Needles and Syringes.
For more information about safe sharps disposal, and for specific information about sharps disposal in the state that you live in, go to the FDA's website at: http://www.fda.gov/safesharpsdisposal
Do not dispose of your used sharps disposal container in your household trash unless your community guidelines permit this. Do not recycle your used sharps disposal container.
Do not put the Needle Cap back on.
This Instructions for Use has been approved by the U.S. Food and Drug Administration.
REGENERON
SANOFI GENZYME
Manufactured by:
Regeneron Pharmaceuticals, Inc.
Tarrytown, NY 10591
U.S. License No. 1760
Marketed by:
sanofi-aventis U.S. LLC (Bridgewater, NJ 08807) and
Regeneron Pharmaceuticals, Inc. (Tarrytown, NY 10591)
DUPIXENT® is a registered trademark of Sanofi Biotechnology
© 2017 Regeneron Pharmaceuticals, Inc. / sanofi-aventis U.S. LLC. All rights reserved.
Issue Date: August 2017
Principal Display Panel - 300 Mg/2 Ml Syringe Carton - Ndc 0024-5914-01
NDC 0024-5914-01
Rx Only
DUPIXENT®
(dupilumab)
Injection
300 mg/2 mL (150 mg/mL)
For Subcutaneous Injection Only.
300 mg/2 mL
2
Single-dose
Pre-filled Syringes
with Needle Shield
Keep out of reach of children. Do not use after
expiration. Do not use if seal is broken or damaged.
Store refrigerated at 36° to 46°F (2° to 8°C) in the original carton.
Dosage and Administration: See package insert for dosage information and directions for use.
SANOFI GENZYME
REGENERON
Principal Display Panel - 300 Mg/2 Ml Syringe Carton - Ndc 0024-5916-01
NDC 0024-5916-01
Rx Only
DUPIXENT®
(dupilumab)
Injection
300 mg/2 mL (150 mg/mL)
For Subcutaneous Injection Only.
300 mg/2 mL
2
Single-dose
Pre-filled Syringes
Keep out of reach of children. Do not use after
expiration. Do not use if seal is broken or damaged.
Store refrigerated at 36° to 46°F (2° to 8°C) in the original carton.
Dosage and Administration: See package insert for dosage information and directions for use.
SANOFI GENZYME
REGENERON
* Please review the disclaimer below.