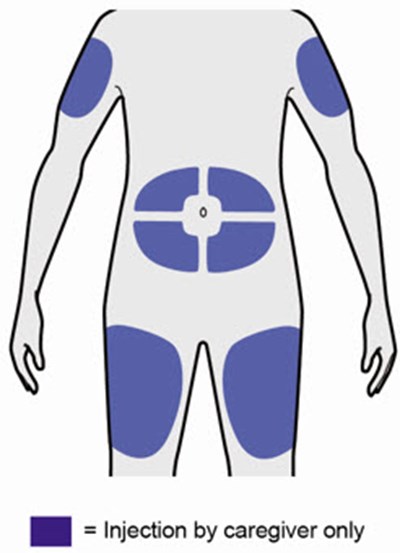
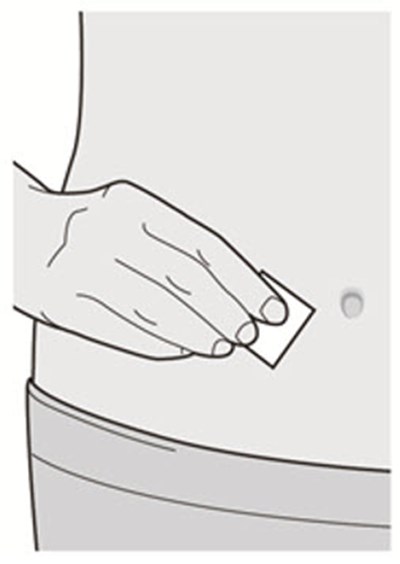
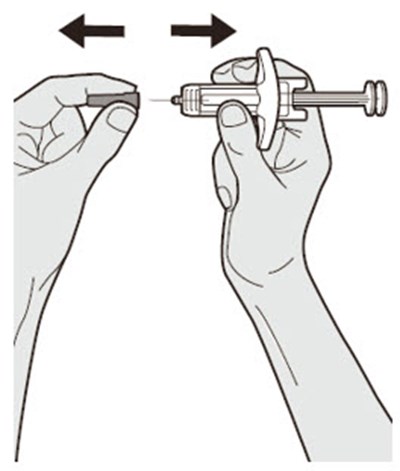
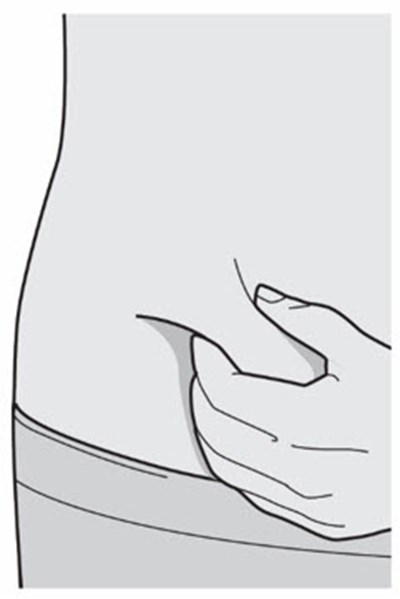
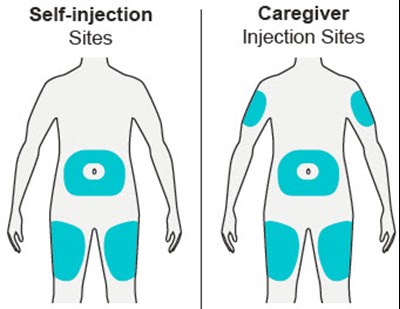
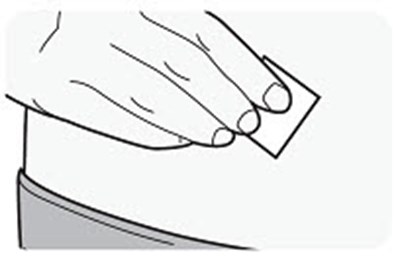
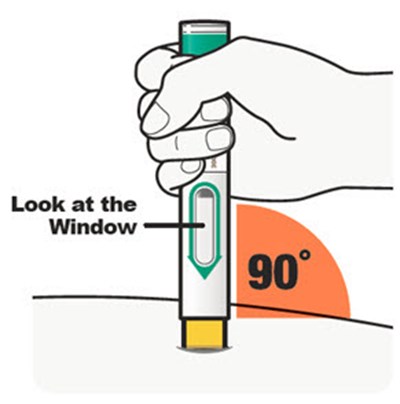
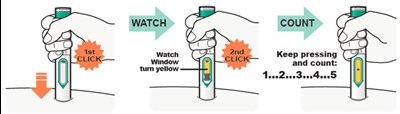
Product Images Dupixent
View Photos of Packaging, Labels & Appearance
- Figure - dupixent 01
- Figure - dupixent 02
- Figure - dupixent 03
- Figure - dupixent 04
- Figure - dupixent 05
- Figure - dupixent 06
- Figure - dupixent 07
- Figure - dupixent 08
- Figure - dupixent 09
- Figure - dupixent 10
- dupixent 100
- dupixent 101
- dupixent 102
- dupixent 103
- dupixent 104
- dupixent 105
- dupixent 106
- dupixent 107
- dupixent 108
- dupixent 109
- Figure - dupixent 11
- dupixent 110
- dupixent 111
- dupixent 112
- dupixent 113
- dupixent 114
- dupixent 115
- dupixent 116
- dupixent 117
- dupixent 118
- dupixent 119
- dupixent 11a
- dupixent 11b
- dupixent 11c
- Figure - dupixent 12
- dupixent 120
- dupixent 121
- dupixent 122
- dupixent 123
- dupixent 124
- dupixent 125
- dupixent 126
- dupixent 127
- dupixent 128
- dupixent 129
- Figure - dupixent 13
- dupixent 130
- Figure - dupixent 14
- Figure - dupixent 15
- Figure - dupixent 16
- Figure - dupixent 17
- Figure - dupixent 18
- PRINCIPAL DISPLAY PANEL - 300 mg/2 mL Syringe Carton - NDC 0024-5914-01 - dupixent 19
- PRINCIPAL DISPLAY PANEL - 300 mg/2 mL Syringe Carton - NDC 0024-5916-01 - dupixent 20
- dupixent 21
- dupixent 22
- dupixent 23
- dupixent 24
- dupixent 25
- dupixent 26
- dupixent 27
- dupixent 28
- dupixent 29
- dupixent 30
- dupixent 31
- dupixent 32
- dupixent 33
- dupixent 34
- dupixent 35
- dupixent 36
- dupixent 37
- dupixent 38
- dupixent 39
- dupixent 40
- dupixent 41
- dupixent 42
- dupixent 43
- dupixent 44
- dupixent 45
- dupixent 46
- dupixent 47
- dupixent 48
- dupixent 49
- dupixent 50
- dupixent 51
- dupixent 52
- dupixent 53
- dupixent 54
- dupixent 55
- dupixent 56
- dupixent 57
- dupixent 58
- dupixent 59
- dupixent 60
- dupixent 61
- dupixent 62
- dupixent 63
- dupixent 64
- dupixent 65
- dupixent 66
Product Label Images
The following 100 images provide visual information about the product associated with Dupixent NDC 0024-5919 by Sanofi-aventis U.s. Llc, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Figure - dupixent 01
The text provided seems to show a graph. Unfortunately, the graph cannot be interpreted with the information solely provided.*
Figure - dupixent 02
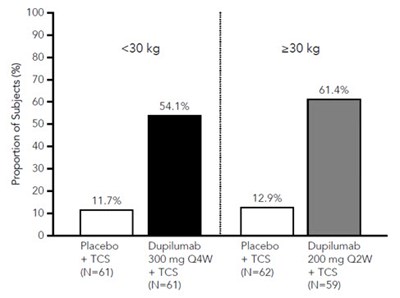
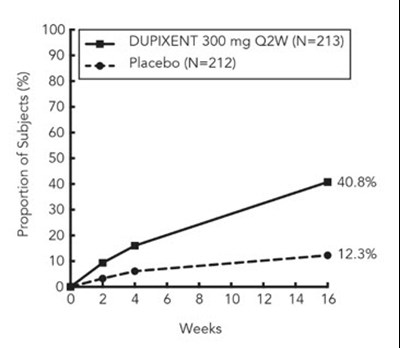
This is a graph showing the proportion of subjects (%) who received either DUPIXENT 300mg Q2W or a placebo over a period of 16 weeks. The proportion of subjects who received DUPIXENT was 36% while the proportion who received a placebo was 5%.*
Figure - dupixent 03
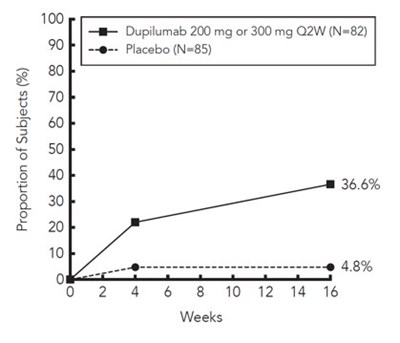
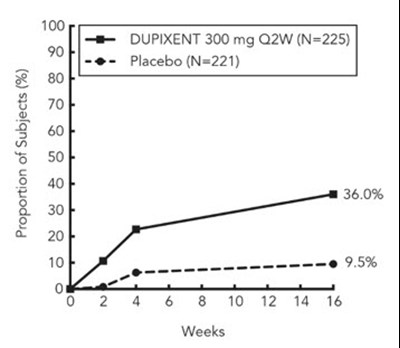
This is a chart presenting the proportion of subjects in a study. The study compares the efficacy of Dupilumab (at doses of 200mg or 300mg every two weeks) to a placebo. The chart shows that after 16 weeks, 36.6% of subjects on Dupilumab experienced a positive outcome.*
Figure - dupixent 05
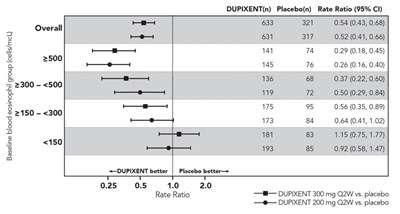
This appears to be a table showing rates and ratios for a drug called Dupixent compared to a placebo. The table includes information on different dosages and categories. However, without additional context or headings, it is difficult to provide a more detailed description of the data presented.*
Figure - dupixent 06
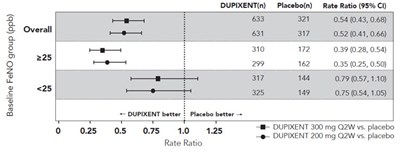
This is a table presenting the baseline FeNO group (ppb) and rate ratio for DUPIXENT compared to a Placebo. The values in the table are not interpretable and require further context and information to be understood.*
Figure - dupixent 07

This appears to be a clinical trial report for a medication called DUPIXENT. The trial involved 277 participants taking a dosage of 300mg every two weeks, 264 participants taking a dosage of 200mg every two weeks, and a placebo group consisting of 142 participants. The report includes a graph showing the efficacy of the medication over time, with measurements taken at various weeks.*
Figure - dupixent 08
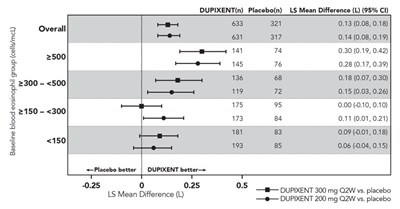
This is a table presenting data from a clinical trial that compared the efficacy of Dupixent (at doses of 200mg Q2W and 300mg Q2W) versus placebo. The numbers in the column indicate the sample size for each group, and the values in the last column represent the mean difference (LS) between Dupixent and the placebo group. The numbers in parenthesis indicate the 95% confidence interval for the mean difference. The table suggests that both doses of Dupixent were more effective than the placebo. However, further information on the nature of the trial and study design is required for a more accurate interpretation.*
Figure - dupixent 09
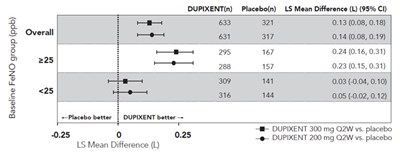
This text is a summary table of a clinical trial that compares the effect of two doses of DUPIXENT and placebo on Baseline FeNO levels in a group of subjects. The results indicate that overall, DUPIXENT is better than the placebo in reducing FeNO levels with statistically significant differences between the LS mean difference of DUPIXENT and the placebo for both doses tested. The LS mean difference between the DUPIXENT doses is not shown.*
Figure - dupixent 10
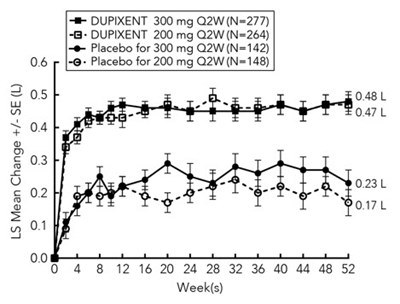
This is a study comparing the effectiveness of a drug called Dupixent at two different doses (300 mg and 200 mg) every two weeks vs placebo in a sample of 733 individuals. The text includes data on the number of participants in each group over the course of 52 weeks, as well as the mean change in an unspecified outcome variable measured at various time points.*
dupixent 101

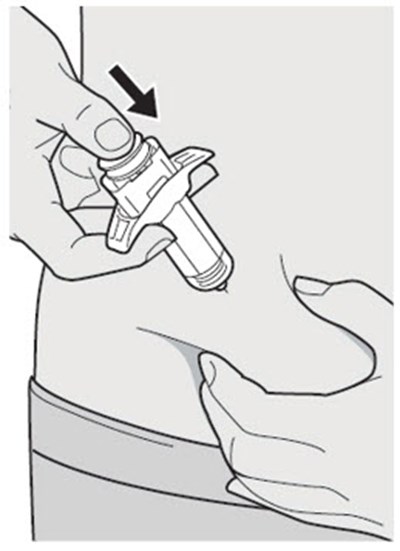
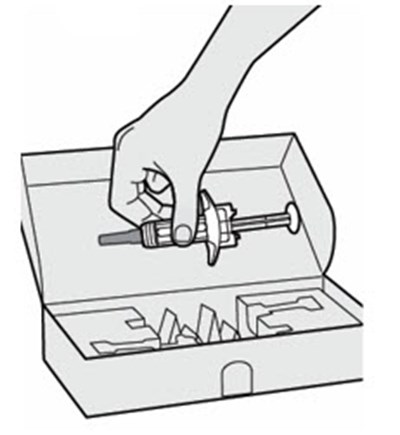
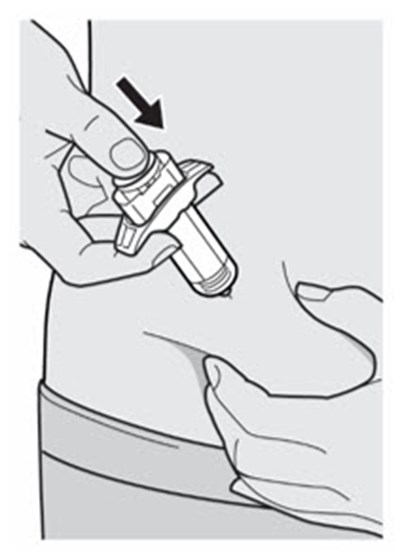
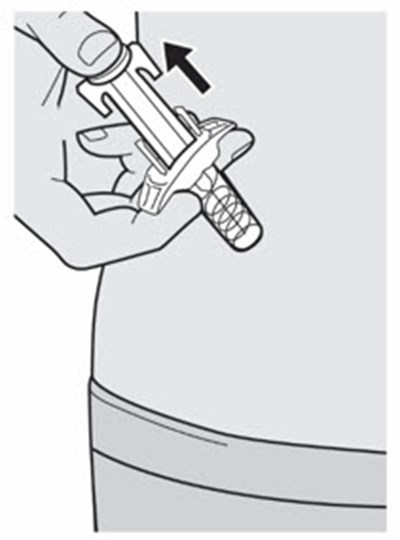
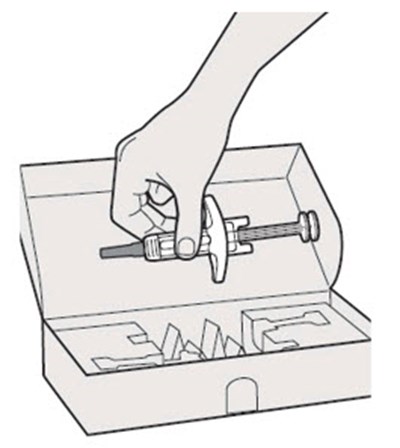
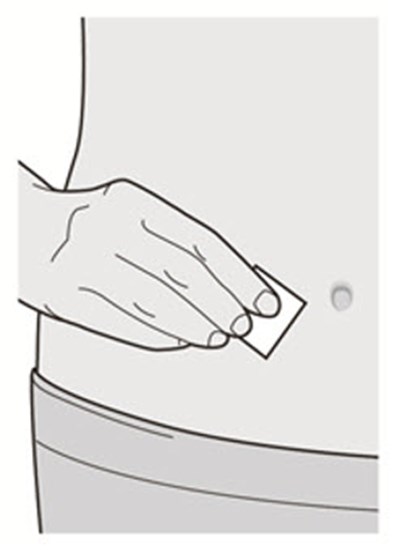
The text describes the contents of a pre-filed syringe of DUPIXENT, which includes one alcohol wipe, one cotton ball or gauze, and a sharps disposal container. DUPIXENT is a medication used to treat certain inflammatory conditions like eczema and asthma.*
Figure - dupixent 11
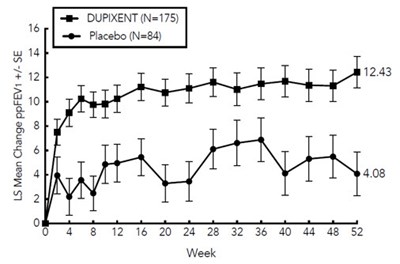
This appears to be a chart or graph showing change in ppFEV1 (a measure of lung function) over time in weeks for a group of 175 people taking Dupixent compared to a group of 84 people taking a placebo. The indicated LS (least squares) mean change is -8 for the Dupixent group. The SE (standard error) values are given as 12.43 at week 0 and 4.08 at week 52.*
dupixent 11a
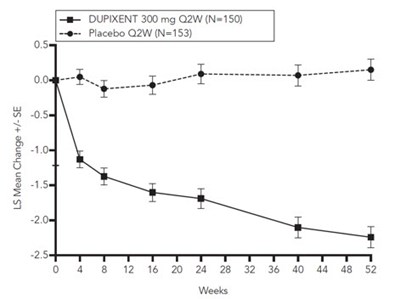
This data appears to be results from a study comparing the medication Dupixent 300 mg to a placebo taken every two weeks. The chart shows the LS Mean Change +/-SE (standard error) over weeks 8 through 40, with possibly some measurements at 0.5, 5.4, and -1.04. It is not clear what the units of measurement or the specific condition being treated are.*
dupixent 11b

This text shows the mean change of DUPIXENT (a medication) compared to a placebo every two weeks over a period of 48 weeks, based on a sample size of 143 and 133 respectively. The mean change is presented in percentages at different time intervals (weeks).*
dupixent 11c
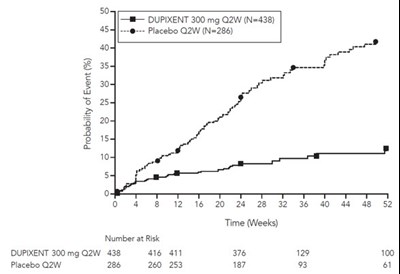
This is a clinical trial report comparing the effect of Dupixent 300 mg Q2W with placebo Q2W on an unknown health condition. The report shows the number of participants in each group (438 in the Dupixent group and 286 in the placebo group) along with their status at different time points (4, 8, 12, 16, 20, 28, 32, 36, 40, 44, 48, 52 weeks). The report also shows the number of participants at risk at each time point.*
dupixent 126

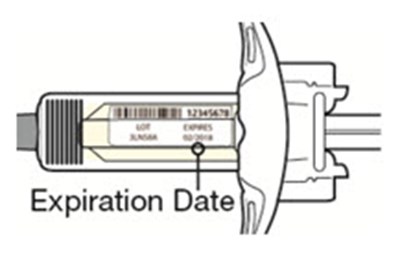
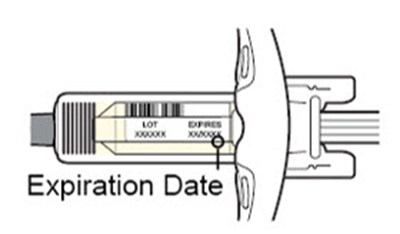
A medication named Dupixent with the NDC code 0024-5914.01 is described. It is an injection given subcutaneously (under the skin). Each pre-filled syringe contains 300mg/2mL of dupilumab. The medication should be stored at a temperature of 2°C to 8°C and must be protected from light. The product must not be frozen. The medication is manufactured by Sanofi Genzyme and Regeneron. Detailed instructions regarding dosage and administration can be found in the package insert.*
dupixent 127

This is a description of a drug called Dupixent, which is administered through subcutaneous injection. The drug comes in a prefilled syringe and each syringe delivers a 200mg dose. The drug must be stored in a refrigerator at a temperature of 36-46°F and protected from light. It should not be used after 14 days or if it has been exposed to direct sunlight. The package insert should be reviewed for dosage and administration information. These details are important to ensure proper use and effectiveness of the drug.*
dupixent 128

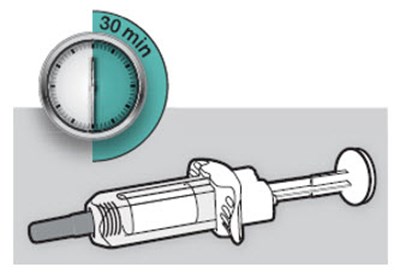
This is a product information label for Dupixent, an injection medication used to treat certain inflammatory conditions such as eczema and asthma. It comes in a single-dose pre-filled pen containing 300mg/2mL of dupilumab, to be administered subcutaneously. The pen also includes several other ingredients, including L-arginine hydrochloride and sucrose. The package includes instructions for use and dosage information. It is important to store the medication at room temperature and avoid exposure to sunlight for up to 14 days. NDC code: 0024-5915-02.*
dupixent 129

This is packaging information for Dupixent, a medication that is stored refrigerated and kept away from heat sources. It is administered via injection and comes in a single-dose, pre-filled pen. The packaging gives instructions for dosing and administration, and includes a warning to keep the medication out of reach of children. The text also includes various identification codes for the product.*
dupixent 130

This is a description of DUPiXENT, a medication that comes in single-dose pre-filled syringes for injection. Each syringe contains 100 mg of dupilumab, which is intended to be kept at room temperature for up to 14 days. The medication should not be exposed to temperatures above 77°F (25°C) and should be discarded after use. The recommended dosage and administration instructions can be found in the package insert, and more information about DUPiXENT can be found at DUPiXENTinfo.com.*
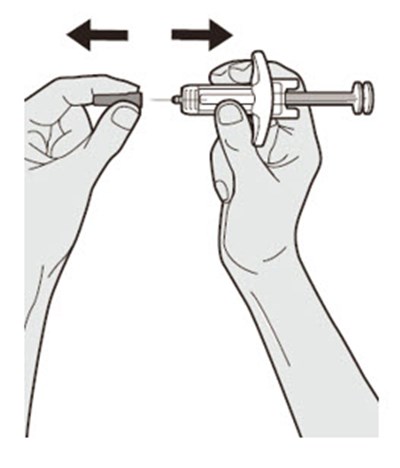
Figure - dupixent 15

This is a pack of DUPIXENT pre-filled syringes along with an alcohol wipe, a cotton ball or gauze and a sharps disposal container for safe disposal of used syringes.*
PRINCIPAL DISPLAY PANEL - 300 mg/2 mL Syringe Carton - NDC 0024-5914-01 - dupixent 19
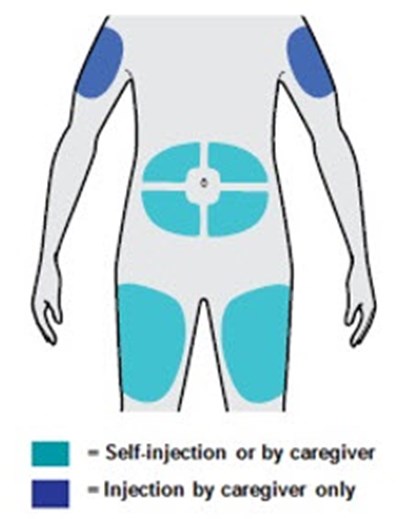
This text is not comprehensive enough to provide a useful description.*
dupixent 30

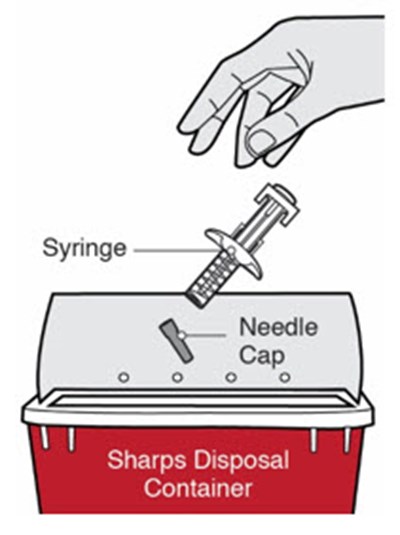
This is a description of the contents of a DUPIXENT pre-filled syringe package. It includes one alcohol wipe and a sharps disposal container.*
dupixent 32
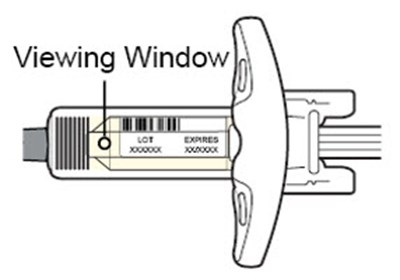
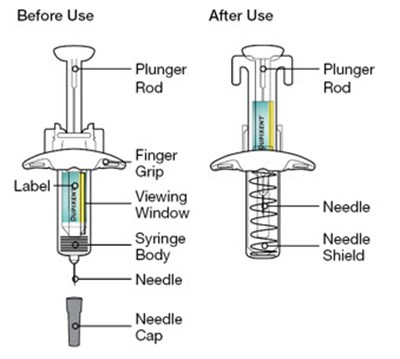
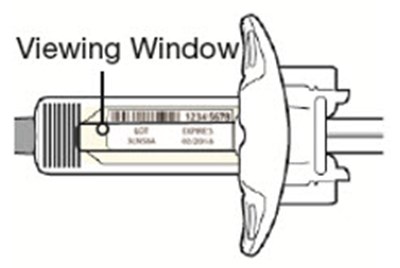
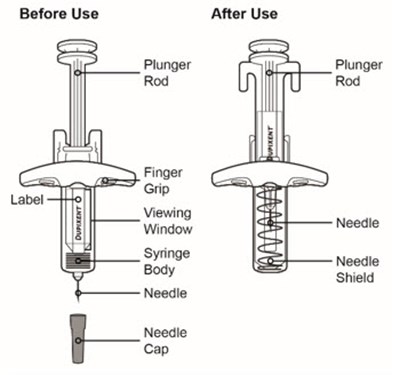
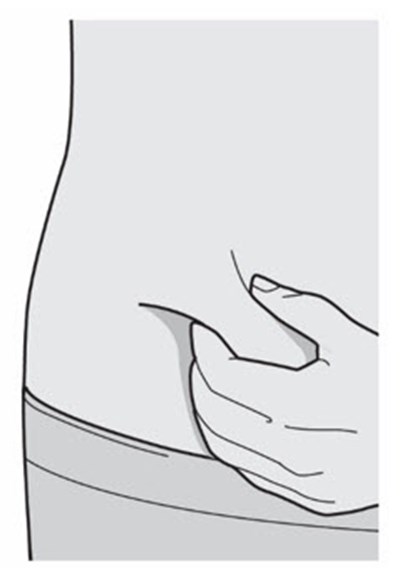
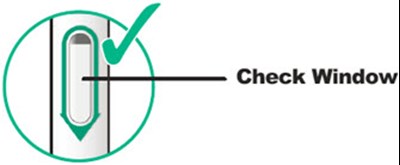
This description is for a product or an element in a user interface that is called "Viewing Window". No further information is provided.*
dupixent 41
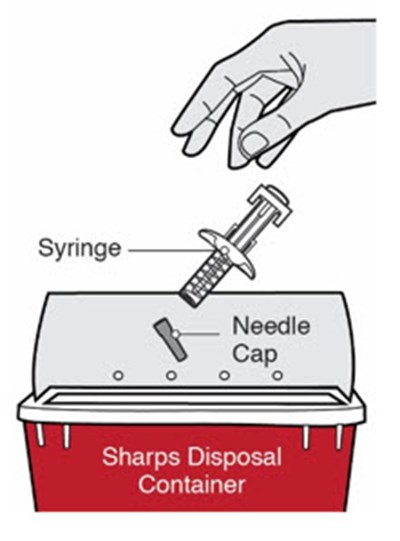
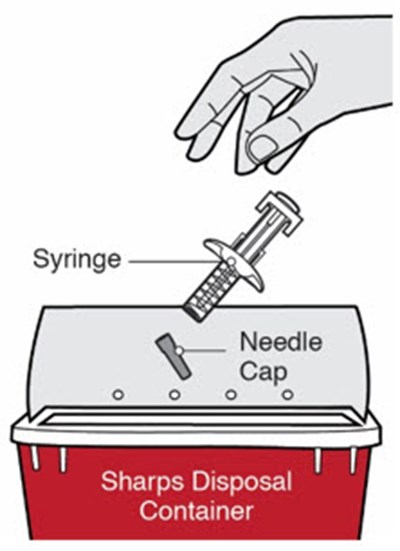
This is a description of a product for maintaining medical hygiene. The product is a container for disposing of used syringes and other sharps.*
dupixent 42
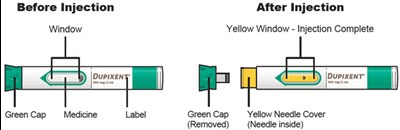
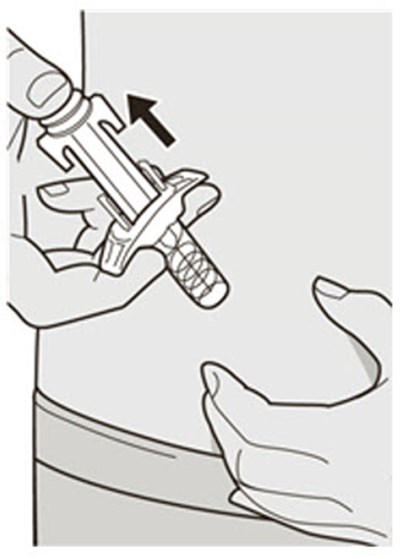
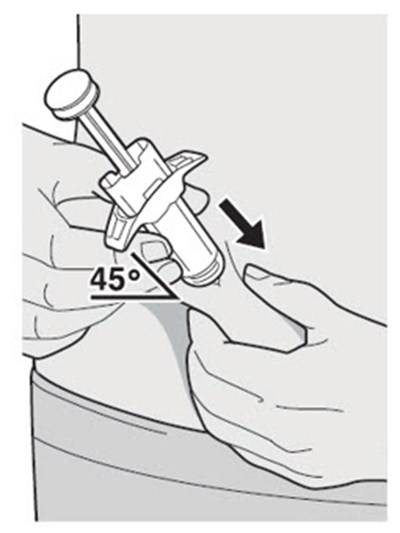
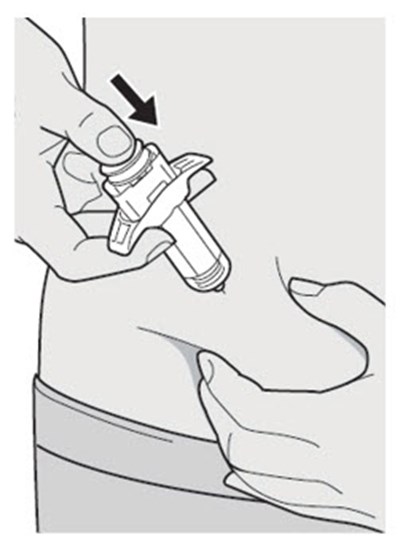
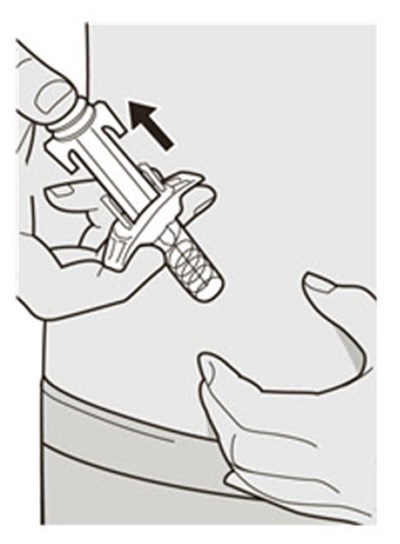
This text appears to be describing an injection process. It shows "Before Injection" and "After Injection" sections with some related details, such as the color of the window changing from yellow to indicate that the injection is complete. Additionally, there are descriptions of various items including a green cap, medicine label, and yellow needle cover. The text does not provide enough information to determine what type of injection, medication, or medical procedure is being performed.*
dupixent 43

The text describes the contents of a package of DUPIXENT pre-filled pen. It includes a cotton ball or gauze, an alcohol wipe and a sharps disposal container. However, these items are not included in the carton.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.