Product Images Irbesartan And Hydrochlorothiazide
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 10 images provide visual information about the product associated with Irbesartan And Hydrochlorothiazide NDC 0054-0254 by Hikma Pharmaceuticals Usa Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Figure1b.jpg - Figure1b
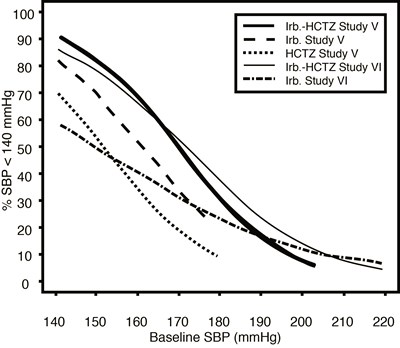
This appears to be a table of blood pressure readings in mmHg from several studies, including Irb.-HCTZ Study V and VI, as well as a HCTZ Study V. The readings range from 100 to 220. There is not enough information provided to determine the purpose or context of this table.*
Figure2a.jpg - Figure2a

This appears to be a list of names of medical studies followed by a chart indicating baseline diastolic blood pressure measurements in mmHg. However, without further context or information, it is not clear what these studies are specifically investigating or what the significance of the blood pressure measurements are.*
Figure2b.jpg - Figure2b

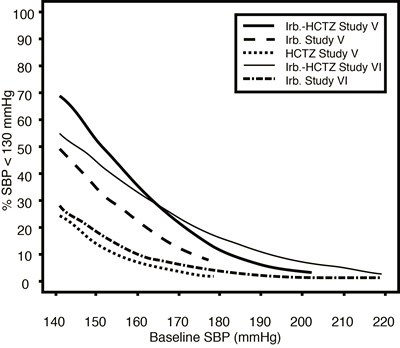
This appears to be a graph showing blood pressure readings for two different studies (Study V and VI) with different medications - one with Irb. (possibly Irbesartan) and one with HCTZ (Hydrochlorothiazide). The bottom axis shows baseline diastolic blood pressure (DBP) measurements in mmHg. The vertical axis shows the percentage of patients with DBP below 80 mmHg. The graph may be interpreting the effectiveness of each medication in reducing DBP below 80mmHg.*
fpl-bl-150mgper12pt5mg-30tab-02.jpg - fpl bl 150mgper12pt5mg 30tab 02

Irbesartan and Hydrochlorothiazide Tablets USP each contain 150mg of irbesartan USP and 12.5 mg of hydrochlorothiazide USP. The usual dosage is available in the package insert for complete prescribing information. It should be stored at 20° to 25°C (68° to 77°F) according to USP controlled room temperature. The tablet is distributed by West-Ward Pharmaceuticals Corp., Eatontown, NJ 07724. The product code is NDC 0054-0254-13 with 30 tablets in each pack.*
fpl-bl-300mgper12pt5mg-30tab-02.jpg - fpl bl 300mgper12pt5mg 30tab 02

This is a description of a medication containing 300 mg of irbesartan USP and 12.5 mg of Irbesartan hydrochlorothiazide USP. It comes in the form of 30 tablets with a National Drug Code (NDC) of 0054-0255-13. The tablets are prescribed for a range of ailments, and it is important to store them at 20° to 25°C or 68° to 77°F. It is distributed by West-Ward Pharmaceuticals Corp. and the product code is 10005169/02.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.