Product Images Everolimus
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 10 images provide visual information about the product associated with Everolimus NDC 0054-0497 by Hikma Pharmaceuticals Usa Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
image 01

Each tablet contains 2.5 g everolimus. The usual dosage should be consulted through the package insert available. The tablets should be stored in their original containers at 20° to 25°C protecting them from light and moisture. The drug must always be kept out of children's reach. The tablets are distributed by Hikma Pharmaceuticals USA Inc. with a strength of 25 mg. A National Drug Code is also provided (NDC 0054-0480-13) as well as a product code (50000030702).*
image 02
This is a description of uni-dose tablets with the active ingredient Everolimus, with each tablet containing 25 mg of the ingredient. The usual dosage is S0 package out for l Tablets. It suggests storing the tablets in a cool and dry place away from light and moisture. The manufacturer is PharmaceuticalsUSA Inc, located in New Jersey.*
image 03
Each tablet contains 5g of the drug Everolimus for oral use. The usual dosage is available in the package insert for complete prescribing information. The drug should be stored at 20° to 25°C (68° to7°F) in a USP controlled room temperature. The drug should be stored in its original container and protected from light and moisture. Furthermore, it should be kept out of the reach of children. The NDC number of the drug is 0084-0481-13. Hikma Pharmaceuticals USA Inc. based in Berkeley Heights, NJ 07922 is the distributor of the drug in the USA. The package contains 30 tablets.*
image 04
This is information about a medication with the NDC code 005048114. Each tablet contains 5mg of Everolimus. The recommended dosage can be found on the package insert. The tablets should be stored at a controlled room temperature and protected from light and moisture. The medication should be kept out of reach of children. It is distributed by Hikma Pharmaceuticals USA Inc.*
image 06
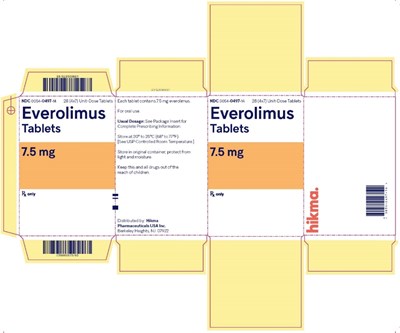
Everolimus is a medication distributed in the form of 28 unit-dose tablets. The prescribing information is available on the package. It should be stored at room temperature and kept away from children. The tablets are distributed by Hitama Pharmaceuticals USA Inc. The NDC number is 001-0897-14.*
image 08
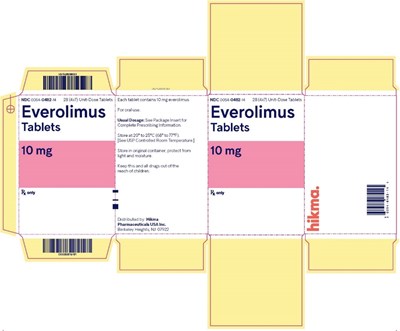
This appears to be a label or packaging of a medication called Everolimus, which comes in tablet form. It includes information such as dosage instructions, storage recommendations, and a warning to keep the drug out of reach of children.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.