Product Images Rufinamide
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 13 images provide visual information about the product associated with Rufinamide NDC 0054-0528 by Hikma Pharmaceuticals Usa Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
rufinamide bottle label

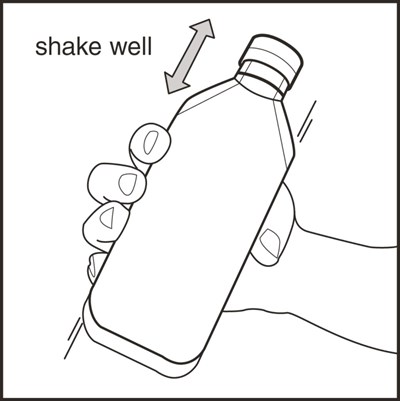
This text provides information about an oral suspension medication called Rufinamide. The NDC code is 0054-0528-63 and it is contained in a 460mL bottle. The medication guide is enclosed and should be dispensed accordingly. The medication is meant for oral administration only and the container should be shaken before each use. It needs to be discarded within 90 days after opening and stored at 20° to 25°C (68° to 77°F), as per USP Controlled Room Temperature guidelines. It is recommended to see the accompanying prescribing information for dosage and use. The manufacturer and distributor of the medication is Hikma Pharmaceuticals USA Inc located in Berkeley Heights, NJ.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.