Product Images Ibrance
View Photos of Packaging, Labels & Appearance
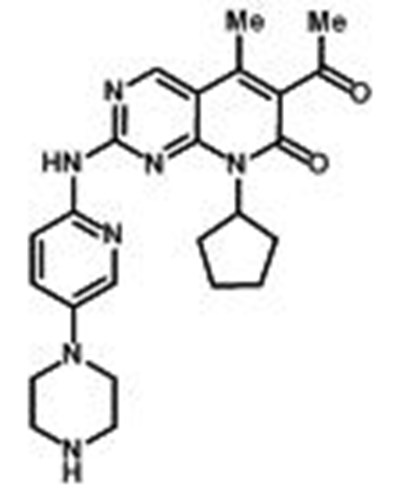
- Chemical Structure - ibrance 01
- Figure 1 - ibrance 02
- Figure 2 - ibrance 03
- Logo - ibrance 04
- PRINCIPAL DISPLAY PANEL - 75 mg Capsule Bottle Label - ibrance 05
- PRINCIPAL DISPLAY PANEL - 100 mg Capsule Bottle Label - ibrance 06
- PRINCIPAL DISPLAY PANEL - 125 mg Capsule Bottle Label - ibrance 07
- ibrance 08
- ibrance 09
Product Label Images
The following 9 images provide visual information about the product associated with Ibrance NDC 0069-0187 by Pfizer Laboratories Div Pfizer Inc, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Figure 1 - ibrance 02
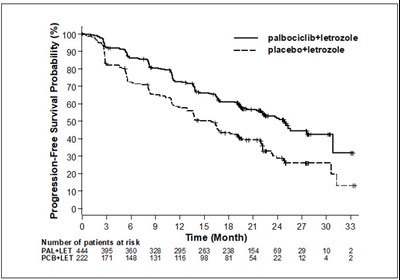
This is a graph depicting the progression-free survival probability percentage of Palbociclib and Letrozole versus Placebo and Letrozole therapy over 33 months. The number of patients at risk is also displayed over time.*
Figure 2 - ibrance 03
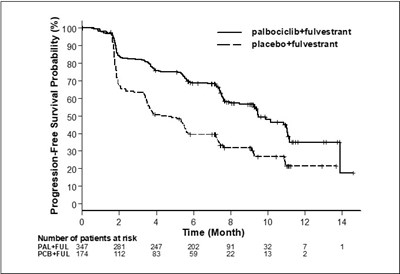
This text shows a chart comparing the Progression-Free Survival Probability between two treatment groups, one being palbociclib+ulvestrant and the other placebo+fulvestrant. It shows the number of patients at risk and the time in months.*
Logo - ibrance 04

This text provides information related to a medical study that involves two treatments: palbociclib+fulvestrant and placebo+fulvestrant. The study records the overall survival probability of the patients over a period of 54 months. The text includes a chart that reports the number of patients at risk at different times of the study for both treatments.*
PRINCIPAL DISPLAY PANEL - 75 mg Capsule Bottle Label - ibrance 05

This is a distribution information of Pfizer Labs that is a division of Pfizer Inc. located in NY, NY 10017.*
PRINCIPAL DISPLAY PANEL - 100 mg Capsule Bottle Label - ibrance 06

This text provides information about the company responsible for distributing a certain product. The product is distributed by Pfizer Labs, which is a division of Pfizer Inc, located in New York City.*
PRINCIPAL DISPLAY PANEL - 125 mg Capsule Bottle Label - ibrance 07

This is a medication information for Ibrance (palbociclib) capsules. It should be stored at a temperature range of 20°C to 25°C (B8°F to 77°F) but can be permitted to store between 15°C to 30°C (59°F to 86°F). It should be dispensed in tight and child-resistant containers. Each capsule contains 75mg of palbociclib, and its distribution is done by Pfizer Labs Division of Pfizer Inc in New York, NY 10017. It is made in Ireland and comes with 21 Capsules. The prescribing information should be consulted for dosage and use. The Global Trade Item Number (GTIN) is 0030069018721, and PAA072956 is a reference number.*
ibrance 08

This is a medication called Ibrance (palbociclib) that comes in capsule form. Each capsule contains 100mg of palbociclb. The medication is distributed by Pfizer Labs in NY and made in Ireland. It should be stored between 20°C to 25°C (B8°F to 77°F) with some permissible temperature excursions between 15°C to 30°C (59°F to 86°F). It should be dispensed in tight, child-resistant containers. Dosage and usage guidance can be found in the accompanying prescribing information. The packaging contains 21 capsules with a Rx only label. The NDC number is 0069-0188-21, and the Bxp and LOT numbers are also provided along with the GTIN number.*
ibrance 09

This is a medication information for Ibrance (palbociclib) capsules distributed by Pfizer Inc. The capsules should be stored at room temperature between 20°C to 25°C but can handle excursions between 15°C to 30°C. The medication should be dispensed in childproof containers. Each capsule holds 125mg of palbociclb. Prescribing information should be consulted before use. The product is manufactured in Ireland, and each package contains 21 capsules. The label also includes a lot number (not readable in the text) and a GTIN number.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.