Product Images Nivestym
View Photos of Packaging, Labels & Appearance
- Logo - nivestym 01
- Logo - nivestym 02
- Figure A - nivestym 03
- Figure B - nivestym 04
- Figure C - nivestym 05
- Figure D - nivestym 06
- Figure E - nivestym 07
- Figure F - nivestym 08
- Figure G - nivestym 09
- Figure H - nivestym 10
- Figure I - nivestym 11
- Figure J - nivestym 12
- Figure K - nivestym 13
- Figure L - nivestym 14
- Figure M - nivestym 15
- Figure N - nivestym 16
- Figure O - nivestym 17
- Figure P - nivestym 18
- Logo - nivestym 19
- Figure - nivestym 20
- Figure - nivestym 21
- Figure - nivestym 22
- Figure - nivestym 23
- Figure - nivestym 24
- Figure - nivestym 25
- Figure - nivestym 26
- Figure - nivestym 27
- Figure - nivestym 28
- Figure - nivestym 29
- Figure - nivestym 30
- Figure - nivestym 31
- Logo - nivestym 32
- PRINCIPAL DISPLAY PANEL - 1 mL Vial Label - nivestym 33
- PRINCIPAL DISPLAY PANEL - 1 mL Vial Carton - nivestym 34
- PRINCIPAL DISPLAY PANEL - 1.6 mL Vial Label - nivestym 35
- PRINCIPAL DISPLAY PANEL - 1.6 mL Vial Carton - nivestym 36
- PRINCIPAL DISPLAY PANEL - 0.5 mL Syringe Label - nivestym 37
- PRINCIPAL DISPLAY PANEL - 0.5 mL Syringe Carton - 71002448 - nivestym 38
- PRINCIPAL DISPLAY PANEL - 0.5 mL Syringe Carton - 10 Pk - nivestym 39
- PRINCIPAL DISPLAY PANEL - 0.5 mL Syringe Carton - 71002449 - nivestym 40
- PRINCIPAL DISPLAY PANEL - 0.8 mL Syringe Label - nivestym 41
- PRINCIPAL DISPLAY PANEL - 0.8 mL Syringe Carton - 71002447 - nivestym 42
- PRINCIPAL DISPLAY PANEL - 0.8 mL Syringe Carton - 10 Pk - nivestym 43
- PRINCIPAL DISPLAY PANEL - 0.8 mL Sryinge Carton - 71002451 - nivestym 44
Product Label Images
The following 44 images provide visual information about the product associated with Nivestym NDC 0069-0292 by Pfizer Laboratories Div Pfizer Inc, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Logo - nivestym 01

The text contains only two fragments of text which are not enough to provide a useful description. The first fragment includes the characters "P zer" and the second fragment includes the word "Oncology". Without further context, it is not possible to generate a description or to infer what the text is referring to.*
Figure B - nivestym 04

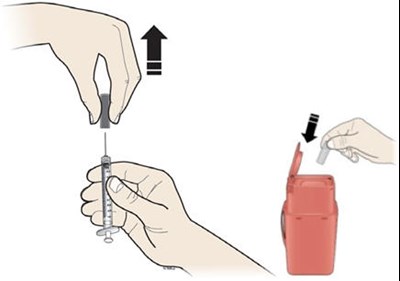
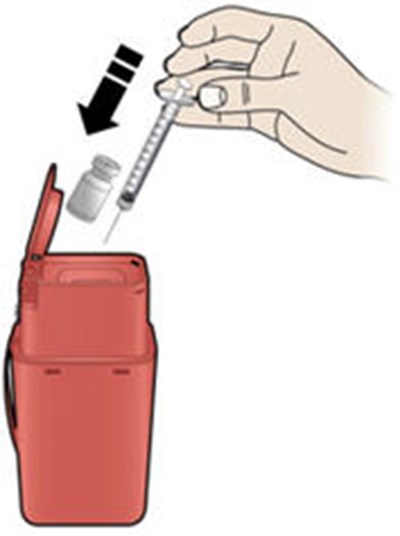
This text describes an alcohol wipe cotton ball, an adhesive bandage, and a sharps disposal container, likely related to medical or first aid use.*
Figure E - nivestym 07
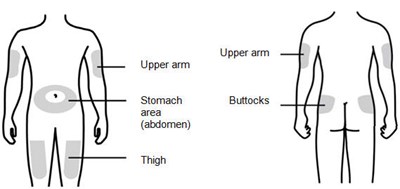
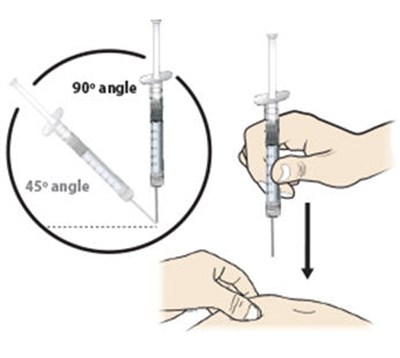
This appears to be a list of body parts including the stomach area, thigh, upper arm, and buttocks.*
PRINCIPAL DISPLAY PANEL - 1 mL Vial Label - nivestym 33

Nivestym is a prescription drug that comes in a 1 mL single-dose vial. It contains filgrastim-aafi and is distributed by Pfizer Labs, a division of Pfizer Inc. It is recommended to store the medication at 2°C to 8°C. The dosage information can be found on the package. The drug is produced by Hospira, Inc. and is a biosimilar. The US License number for this medication is 1974.*
PRINCIPAL DISPLAY PANEL - 1 mL Vial Carton - nivestym 34

Nivestym is a medication that comes in the form of an injection with no preservatives. The provided text also contains various identification numbers, possibly for inventory or tracking purposes, and some illegible characters.*
PRINCIPAL DISPLAY PANEL - 1.6 mL Vial Label - nivestym 35

This is a medication called Nivestym (filgrastim-aafi) Injection that is manufactured by Hospira, Inc. The dosage instructions are on the package. It is important to store it at 2°08°C. The medication comes in a 1.6 mL single-dose vial and is distributed by Pfizer Labs. This medication is a biosimilar. The text includes a reference number of RL10065.*
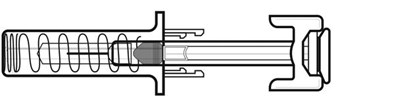
PRINCIPAL DISPLAY PANEL - 0.5 mL Syringe Label - nivestym 37

This text describes a medication called 'filgrastim-aafi' which is sold as a single dose in a syringe. It is for subcutaneous use only and produced by the company 'Hospialnc'. The medication has a concentration of 300mg/0.5mL and comes in a BTV container. The text includes a license number and a production date but does not include any information on the medical uses or side effects of the medication.*
PRINCIPAL DISPLAY PANEL - 0.5 mL Syringe Carton - 71002449 - nivestym 40

This appears to be a product description for a single-dose prefilled syringe with a needle guard. The syringe contains a sterile solution with recombinant Granulocyte Colony Stimulating factor (G-CSF) derived from E. coli. It is for subcutaneous use only and should be discarded if unused. The product is called ivestym™ (filgrastim-aafi) Injection and comes in a 300 mogh.5ml dose.*
PRINCIPAL DISPLAY PANEL - 0.8 mL Syringe Label - nivestym 41

This appears to be the label or packaging information for a medication called "Neulastim" or "Filgrastim-aafi" for subcutaneous use only. It includes the manufacturer's name and license number, as well as production and expiration information, but does not provide information on the medication's purpose or effects.*
PRINCIPAL DISPLAY PANEL - 0.8 mL Syringe Carton - 10 Pk - nivestym 43
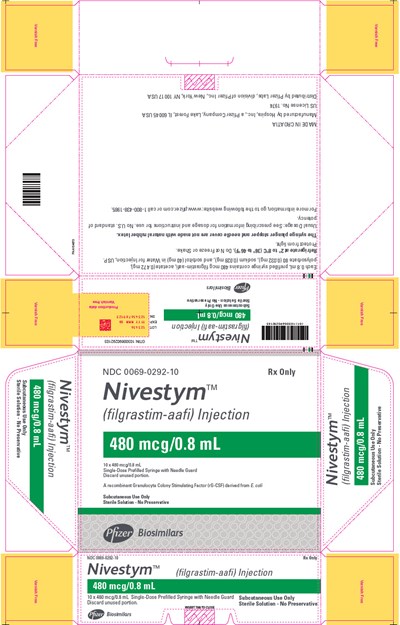
Nivestym is a sterile solution injection of 480 mcg/0.8 mL Filgrastim-aafi, a recombinant Granulocyte Colony Stimulating Factor derived from E. coli, for subcutaneous use only. It is a single-dose prefilled syringe with needle guard. The unused portion should be discarded, and the solution has no preservative.*
PRINCIPAL DISPLAY PANEL - 0.8 mL Sryinge Carton - 71002451 - nivestym 44

This appears to be a mixture of outputs and does not make a coherent description. The text does not appear to be readable and may need to be rescanned or checked for any errors during transcription.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.