Product Images Ngenla
View Photos of Packaging, Labels & Appearance
- Logo - somatrogon 01
- Logo - somatrogon 02
- Logo - somatrogon 03
- Image - somatrogon 05
- Image - somatrogon 06
- Step 2 - somatrogon 07
- Step 3 - somatrogon 08
- Step 4 - somatrogon 09
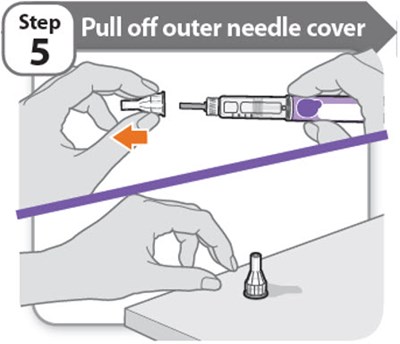
- Step 5 - somatrogon 10
- Step 6 - somatrogon 11
- Image - somatrogon 12
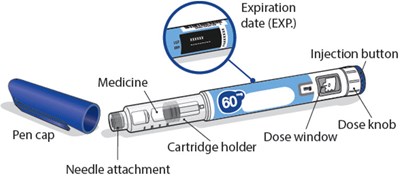
- Image - somatrogon 13
- Image - somatrogon 14
- Image - somatrogon 15
- Step 7 - somatrogon 16
- Example A - somatrogon 17
- Example B - somatrogon 18
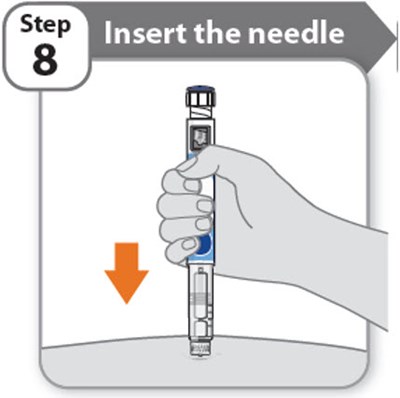
- Step 8 - somatrogon 19
- Step 9 - somatrogon 20
- Step 10 - somatrogon 21
- Step 11 - somatrogon 22
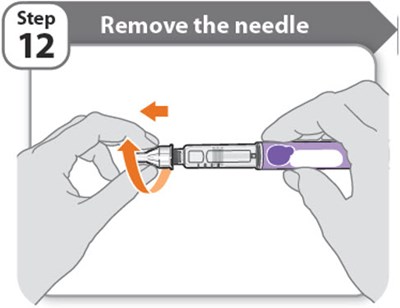
- Step 12 - somatrogon 23
- Step 13 - somatrogon 24
- Logo - somatrogon 25
- Image - somatrogon 26
- Image - somatrogon 27
- Step 2 - somatrogon 28
- Step 3 - somatrogon 29
- Step 4 - somatrogon 30
- Step 5 - somatrogon 31
- Step 6 - somatrogon 32
- Image - somatrogon 33
- Image - somatrogon 34
- Image - somatrogon 35
- Image - somatrogon 36
- Step 7 - somatrogon 37
- Example A - somatrogon 38
- Example B - somatrogon 39
- Step 8 - somatrogon 40
- Step 9 - somatrogon 41
- Step 10 - somatrogon 42
- Step 11 - somatrogon 43
- Step 12 - somatrogon 44
- Step 13 - somatrogon 45
- PRINCIPAL DISPLAY PANEL - 24 mg Cartridge Label - somatrogon 47
- PRINCIPAL DISPLAY PANEL - 24 mg Cartridge Carton - somatrogon 48
- PRINCIPAL DISPLAY PANEL - 60 mg Cartridge Label - somatrogon 49
- PRINCIPAL DISPLAY PANEL - 60 mg Cartridge Carton - somatrogon 50
Product Label Images
The following 48 images provide visual information about the product associated with Ngenla NDC 0069-0505 by Pfizer Laboratories Div Pfizer Inc, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Logo - somatrogon 01

Pfizer Labs, a division of Pfizer Inc., is a distributor of various products. Based in New York, NY 10001, Pfizer Labs offers a range of pharmaceutical and healthcare solutions.*
Logo - somatrogon 02

Distributed by Pfizer Labs, a division of Pfizer Inc., located in New York, NY 10001.*
Logo - somatrogon 03

Distributed by Pfizer Labs, a division of Pfizer Inc., located in New York, NY 10001.*
Step 2 - somatrogon 07

Choose and clean your injection site. For abdominal sites, keep at least 2 inches (5 cm) away from the belly button. For rear upper arms, only caregivers can administer the injection. For front upper thighs, choose this site for injection. For buttocks, only caregivers can administer the injection.*
Logo - somatrogon 25

Distributed by Pfizer Labs, a division of Pfizer Inc. Located in New York, NY 10001.*
Step 2 - somatrogon 28

Choose and clean your injection site. For abdomen sites, keep at least 2 inches (5 cm) away from the belly button. Arms (rear upper) are for caregiver administration only. Thighs (front upper) and buttocks are also for caregiver administration only.*
PRINCIPAL DISPLAY PANEL - 24 mg Cartridge Label - somatrogon 47

Manufactured by Pfizer Ireland Pharmaceuticals in Ringaskiddy, Cork, Ireland. U.S. License No. 2060. This is a medication that is administered through subcutaneous injection. It is a single-patient-use product that should be discarded after 28 days of first use. The medication is stored refrigerated. The specific dosage and concentration are not available due to the unclear text.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.