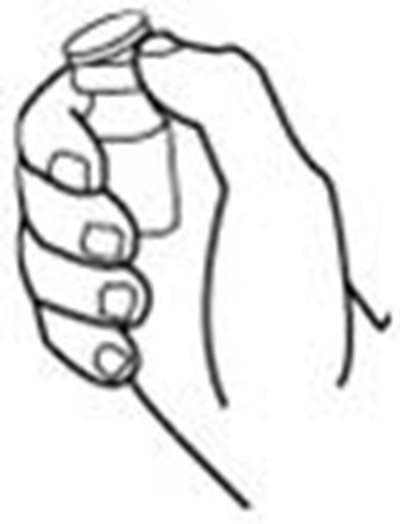
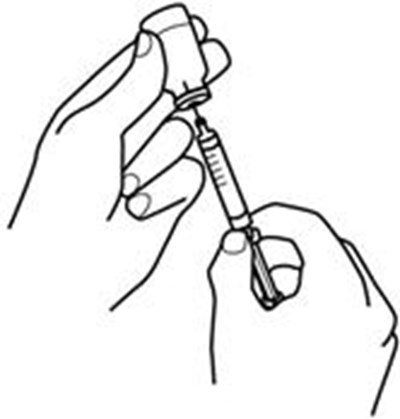
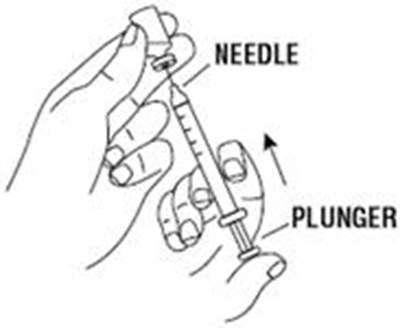
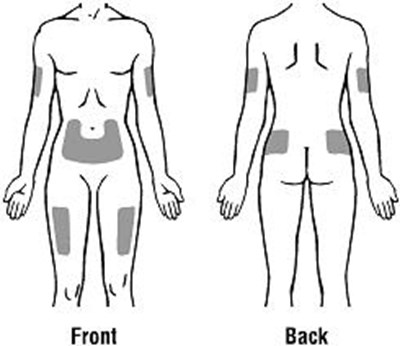
Product Images Retacrit
View Photos of Packaging, Labels & Appearance
- Logo - retacrit 01
- Logo - retacrit 02
- Figure 1 - retacrit 03
- Figure 2 - retacrit 04
- Figure 3 - retacrit 05
- Figure 4 - retacrit 06
- Figure 5 - retacrit 07
- Figure 6 - retacrit 08
- Figure 7 - retacrit 09
- Figure 8 - retacrit 10
- Figure 9 - retacrit 11
- Figure 10 - retacrit 12
- Figure 11 - retacrit 13
- Figure 12 - retacrit 14
- Figure 13 - retacrit 15
- Figure 14 - retacrit 16
- Figure 15 - retacrit 17
- Figure 16 - retacrit 18
- Logo - retacrit 19
- PRINCIPAL DISPLAY PANEL - 2,000 Units/mL Vial Label - retacrit 20
- PRINCIPAL DISPLAY PANEL - 2,000 Units/mL Vial Carton - retacrit 21
- PRINCIPAL DISPLAY PANEL - 3,000 Units/mL Vial Label - retacrit 22
- PRINCIPAL DISPLAY PANEL - 3,000 Units/mL Vial Carton - retacrit 23
- PRINCIPAL DISPLAY PANEL - 4,000 Units/mL Vial Label - retacrit 24
- PRINCIPAL DISPLAY PANEL - 4,000 Units/mL Vial Carton - retacrit 25
- PRINCIPAL DISPLAY PANEL - 10,000 Units/mL Vial Label - retacrit 26
- PRINCIPAL DISPLAY PANEL - 10,000 Units/mL Vial Carton - retacrit 27
- PRINCIPAL DISPLAY PANEL - 40,000 Units/mL Vial Label - retacrit 28
- PRINCIPAL DISPLAY PANEL - 40,000 Units/mL Vial Carton - retacrit 29
- PRINCIPAL DISPLAY PANEL - 20,000 Units/mL Vial Label - retacrit 30
- PRINCIPAL DISPLAY PANEL - 20,000 Units/mL Vial Carton - retacrit 31
- PRINCIPAL DISPLAY PANEL - 20,000 Units/2 mL Vial Label - retacrit 32
- PRINCIPAL DISPLAY PANEL - 20,000 Units/2 mL Vial Carton - retacrit 33
Product Label Images
The following 33 images provide visual information about the product associated with Retacrit NDC 0069-1311 by Pfizer Laboratories Div Pfizer Inc, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
PRINCIPAL DISPLAY PANEL - 2,000 Units/mL Vial Label - retacrit 20

Retacrit® is a medication available in a 2,000 Units/mL single-dose vial, containing epoetin alfa. It is prescribed by healthcare professionals and distributed by Pfizer Labs in New York, NY. The medication should be stored at 2°C to 8°C. Dosage information is available in the prescribing information. The medication is manufactured by Hospira, Inc., a Pfizer Company, and operations are licensed within the USA with License No. 1974.*
PRINCIPAL DISPLAY PANEL - 2,000 Units/mL Vial Carton - retacrit 21

Retacrit® is a solution that contains Epoetin Alfa-epbx recombinant for intravenous or subcutaneous use. It is available in a single-dose vial, and any unused portion should be discarded. The solution is a buffered solution of calcium chloride dihydrate, sodium chloride, and other additives. The vial must be stored refrigerated at 2°C to 8°C. Retacrit® is distributed by Pfizer Labs and should always be dispensed with enclosed medication instructions.*
PRINCIPAL DISPLAY PANEL - 3,000 Units/mL Vial Label - retacrit 22

Retacrit is a medication produced by Hospira, now owned by Pfizer. It is only available through prescription and contains Epoetin Alfa-epbx. The concentration is 3,000 Units/mL and comes in a single-dose vial. It is distributed by Pfizer Labs, which is a division of Pfizer Inc. The medication is used in the field of oncology.*
PRINCIPAL DISPLAY PANEL - 3,000 Units/mL Vial Carton - retacrit 23

Retacrit® is a medication that comes in a single-dose vial containing 1053000 Units of Epoetin Alfa-epbx intended for intravenous or subcutaneous use only. It also contains glycine, isoleucine, leucine, and other components. The medication should not be frozen or shaken, and any unused portion must be discarded. The solution is sterile and does not contain any preservatives. The medication is distributed by Pfizer Labs and is manufactured in Lake Forest, IL, USA.*
PRINCIPAL DISPLAY PANEL - 4,000 Units/mL Vial Label - retacrit 24

NDC 0069-1307-01 is a prescription drug called Retacrit. It contains Epoetin Alfa-epbx and is manufactured by Hospira, Inc, a Pfizer company. The drug is a recombinant protein used to treat anemia. The dosage information can be found on the prescription label. The drug is available in a single-dose vial containing 4,000 Units/mL. It is distributed by Pfizer Labs.*
PRINCIPAL DISPLAY PANEL - 4,000 Units/mL Vial Carton - retacrit 25
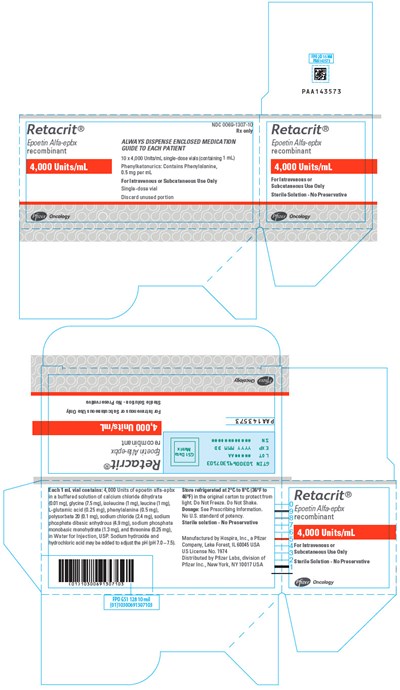
Retacrit® is a recombinant epoetin alfa-epbx solution used for Subcutaneous or Intravenous use. The medication comes in a Single-dose vial and should be refrigerated but not frozen or shaken. It is always recommended to dispense the enclosed medication guide to each patient. Please note that the text provided contains errors and some information is missing.*
PRINCIPAL DISPLAY PANEL - 10,000 Units/mL Vial Label - retacrit 26

Retacrit is a medication that requires a prescription and should be stored at a temperature between 2 and 8 degrees Celsius. It is a Pfizer Inc. product that contains recombinant epoetin alfa and is manufactured by Hospira, Inc. The medication is distributed by Pfizer Labs in the United States under license number 1974. The provided reference number is PAA143574.*
PRINCIPAL DISPLAY PANEL - 10,000 Units/mL Vial Carton - retacrit 27

Retacrit® is a 10,000 Units/mL recombinant solution of epoetin alfa-epbx that can be used for intravenous or subcutaneous delivery. It is a single-dose vial that does not contain any preservatives. Individuals with phenylketonuria should be aware that Retacrit® contains phenylalanine. The medication should be discarded if not used in its entirety, and it should not be shaken before use. The prescribing information should be consulted for proper usage. The solution is manufactured by Hospira, Inc., a Pfizer Company, and distributed by Pfizer Labs.*
PRINCIPAL DISPLAY PANEL - 40,000 Units/mL Vial Label - retacrit 28

This is a medication used for oncology in a single-dose vial with a concentration of 40,000 units per milliliter. The dosage information can be found in the prescribing information. It should be stored at 2°C to 8°C and manufactured by Hospira, a Pfizer company. The distribution of this medication is handled by Pfizer Labs in New York. The lot number and expiration date are provided, but there is no information about what the medication actually does or what it is used for.*
PRINCIPAL DISPLAY PANEL - 40,000 Units/mL Vial Carton - retacrit 29

This is a description of a drug named "Retacrit®", which comes in single-dose vials containing 1 ml of recombinant Epoetin Alfa-epb. The drug is for subcutaneous use only and must be discarded after a single-use. The package includes an enclosed navigation guide to each patient. The text also includes some cautionary notes, a warning for Phenylketonurics and the manufacturing details.*
PRINCIPAL DISPLAY PANEL - 20,000 Units/mL Vial Label - retacrit 30

This is a label of medication produced by Hospira, Inc. which is a subsidiary of Pfizer Inc. The medication is prescribed for Sos (unknown medical condition) and contains important information. The manufacturer's address is located in Lake Forest, IL, and it holds US License No. 1974. The distribution of this medication is by Pfizer Labs, a division of Pfizer Inc located in New York, NY. The number PAA15870 may be a batch number or a specific identifier for this medication.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.