Product Images Doxorubicin Hydrochloride
View Photos of Packaging, Labels & Appearance
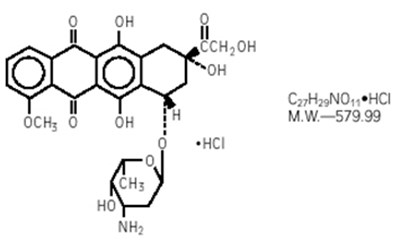
- Chemical Structure - doxorubicin 01
- Figure 1 - doxorubicin 02
- Figure 2 - doxorubicin 03
- Logo - doxorubicin 04
- Logo - doxorubicin 04a
- PRINCIPAL DISPLAY PANEL - 10 mg/5 mL Vial Label - doxorubicin 05
- PRINCIPAL DISPLAY PANEL - 10 mg/5 mL Vial Carton - doxorubicin 06
- PRINCIPAL DISPLAY PANEL - 20 mg/10 mL Vial Label - doxorubicin 07
- PRINCIPAL DISPLAY PANEL - 20 mg/10 mL Vial Carton - doxorubicin 08
- PRINCIPAL DISPLAY PANEL - 50 mg/25 mL Vial Label - doxorubicin 09
- PRINCIPAL DISPLAY PANEL - 50 mg/25 mL Vial Carton - doxorubicin 10
- PRINCIPAL DISPLAY PANEL - 150 mg/75 mL Vial Label - doxorubicin 11
- PRINCIPAL DISPLAY PANEL - 150 mg/75 mL Vial Carton - doxorubicin 12
- PRINCIPAL DISPLAY PANEL - 200 mg/100 mL Vial Label - doxorubicin 13
- PRINCIPAL DISPLAY PANEL - 200 mg/100 mL Vial Carton - doxorubicin 14
Product Label Images
The following 15 images provide visual information about the product associated with Doxorubicin Hydrochloride NDC 0069-3033 by Pfizer Laboratories Div Pfizer Inc, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Figure 1 - doxorubicin 02
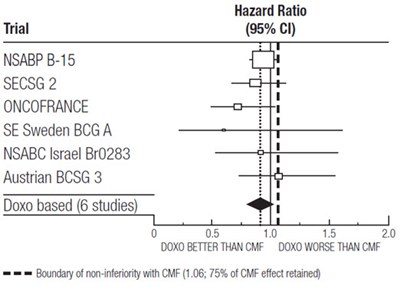
This text appears to be describing various clinical trials and their respective hazard ratios. It also mentions a comparison between treatment with Doxo (a chemotherapy drug) versus CMF (a combination of chemotherapy drugs) and includes a graph showing the superiority and inferiority of Doxo compared to CMF. The text also includes a boundary for non-inferiority with CMF at 75% of the CMF effect retained.*
Figure 2 - doxorubicin 03
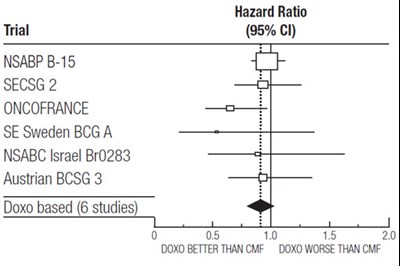
This appears to be a comparison of different cancer treatments based on their hazard ratio in various clinical trials. The text lists the names of the trials and studies with their respective hazard ratios and a comparison between Doxo (Doxorubicin) based treatment and CMF (cyclophosphamide, methotrexate, fluorouracil) treatment. The table suggests that in the six studies listed, Doxo was found to be better than CMF in four of them, and worse than CMF in two.*
Logo - doxorubicin 04

This text is a label mentioning the name of a pharma company "Distributed by Plizer Labs" and a product category "Injectables". The product is under the division of Pfizer Inc located in New York.*
Logo - doxorubicin 04a

The text is a distribution information for injectables by Plizer Labs, a division of Pfizer Inc located in New York, NY 10017.*
PRINCIPAL DISPLAY PANEL - 10 mg/5 mL Vial Label - doxorubicin 05

This text belongs to a prescription medication with the NDC number 0069-3030-20. It is a single-dose Cytosafe Vial that contains a sterile and isotonic solution of Doxorubicin Hydrochloride Injection, with a dose of 10 mg/5mL. This medication is intended for intravenous use only as it is a cytotoxic agent. The vial needs to be stored between 2°C and 8°C (36°F and 46°F), and any unused portion should be discarded. It is important to protect the medication from light. Further dosage and usage instructions can be found in the accompanying prescribing information. This medication has been manufactured in Australia.*
PRINCIPAL DISPLAY PANEL - 10 mg/5 mL Vial Carton - doxorubicin 06

This is a description of a drug called "DOXOrubicin Hydrochloride Injection" used for intravenous use that comes in a "Cytosafe Vial". It should be stored in a cool place between 2-46F and protected from light until use. Each milliliter of the injection contains 2mg of Gocorubicin hyclate, which is equivalent to 1.67mg of Doxobicin, and 50mg of hydrochloric acid. The dosage and prescription are provided by the prescribing physician. It is made in Australia and is a cytotoxic agent. The label says, "Rxonly". The text is difficult to read due to , but this is a rough approximation.*
PRINCIPAL DISPLAY PANEL - 20 mg/10 mL Vial Label - doxorubicin 07

This is a description of a medication with the National Drug Code (NDC) 0069-3031-20 which is a sterile, isotonic solution. The medication is only for intravenous use and is a cytotoxic agent made in Australia with caution. It is a single-dose vial called Gytosafe® and should be discarded if unused. The medication needs to be stored refrigerated between 2 and 8 degrees Celsius and protected from light. The solution contains DOXOrubicin Hydrochloride Injection at a concentration of 20 mg/10mL and the prescribing information should be consulted for dosage and use. The LOT and EXP numbers are also provided.*
PRINCIPAL DISPLAY PANEL - 20 mg/10 mL Vial Carton - doxorubicin 08

This is a description of a drug called "Ster, lotoni Solution". The drug contains 2mg of doxorubicin hydrochloride, which is equivalent to 101.87 mg of B and 9 mg of sodium chloride. The solution has a pH of 3.0 and is administered through intravenous injection. It is recommended to store the drug in a cool place between 2 and 8 degrees Celsius and protect it from light. The dosage and usage information are not available in the text. The drug is available in a single-use Cytosafe vial, and caution is advised as it is a cytotoxic agent. The remaining text is not readable.*
PRINCIPAL DISPLAY PANEL - 50 mg/25 mL Vial Label - doxorubicin 09

This is a description for a pharmaceutical product. The product is a single-dose sterile isotonic solution presented in a Cytosafe Vial. The product is only available with a prescription, and the unused portion should be discarded. The product needs to be stored refrigerated between 2° and 8°C. The product is intended for intravenous use only, and relevant dosage and use information should be found in g2 of Hydrochloride Injection information. The product is made in Australia and contains 50 mg/25 mL, with a strength of 2 mg/mL. A caution is indicated due to it being a Cyotoc Agont QD Injectable.*
PRINCIPAL DISPLAY PANEL - 50 mg/25 mL Vial Carton - doxorubicin 10
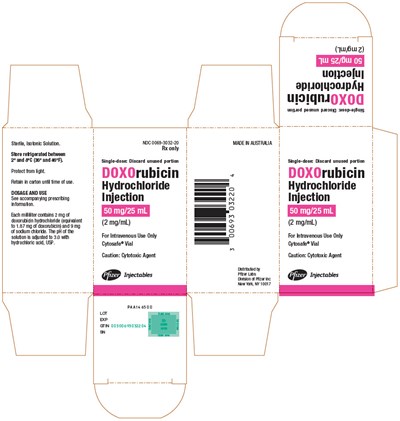
This is a pharmaceutical product that contains doxorubicin hydrochloride used as a Cytotoxic Agent for Intravenous use only. The solution is contained in a Cytosafe Vial, which has to be refrigerated between 28 and 6 degrees Fahrenheit. Each milliliter of the solution contains 2 mg of doxorubicin hydrochloride (equal to 101.67 mg of doxombhin) and 5 mg of sodium chloride. The prescribing information should be consulted before use, and the unused portion should be discarded after a single use. NDC 0069-303220 is the drug code or identifier.*
PRINCIPAL DISPLAY PANEL - 150 mg/75 mL Vial Label - doxorubicin 11

This is a description of Doxorubicin Hydrochloride Injection, a cytotoxic agent, intended for intravenous use only. It is available in multiple-dose vial R, with each milliliter containing 2 mg of doxorubicin hydrochloride and 9 mg of sodium chloride. The solution is isotonic and sterile, packed in a Cytosafe vial. It is recommended to store it between 2°C and 8°C and protect it from light. The Pfizer Labs in New York distribute it, and it is made in Australia. More information is available in the prescribing information.*
PRINCIPAL DISPLAY PANEL - 200 mg/100 mL Vial Label - doxorubicin 13

This is a description of a medication product called NDC 0069-3034-20. It is a sterile, isotonic solution in a multiple-dose vial that requires refrigeration and is only available via prescription. The solution contains DOXOrubicin hydrochloride for intravenous use only with each milliliter containing 2 mg of the drug and 9 mg of sodium chloride. The pH of the solution is adjusted to 3 using hydrochloric acid, USP. It is a cytotoxic agent and must be protected from light. The text appears to be an extract from the drug's prescribing information. The product is made in Australia.*
PRINCIPAL DISPLAY PANEL - 200 mg/100 mL Vial Carton - doxorubicin 14

This appears to be a cautionary label for a cytotoxic agent called DOXOrubicin Hydrochloride. The label indicates that it should be stored refrigerated between 29 and 8°C, and comes in a multiple-dose vial. The label also indicates that it is manufactured by PleerLibs, a division of Przer g in New York. The text further warns that this is a cytotoxic agent and should be used with caution.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.