Product Images Zithromax
View Photos of Packaging, Labels & Appearance
- Chemical Structure - zithromax 01
- Logo - zithromax 02
- zithromax 02a
- PRINCIPAL DISPLAY PANEL - 250 mg - 30 Tablet Bottle Label - zithromax 03
- Principal Display Panel - 250 mg - 6 ct. Blister Pack - zithromax 04
- Principal Display Panel - 250 mg - 10 ct. Blister Pack - zithromax 05
- PRINCIPAL DISPLAY PANEL - 250 mg - 18 Tablet Blister Pack - zithromax 06
- PRINCIPAL DISPLAY PANEL - 250 mg Tablet Blister Pack Box - zithromax 07
- PRINCIPAL DISPLAY PANEL - 500 mg - 30 Tablet Bottle Label - zithromax 08
- Principal Display Panel - 500 mg - 3 ct. Tablet Blister Pack - zithromax 09
- Principal Display Panel - 500 mg - 3 ct. Tablet Blister Carton - zithromax 10
- PRINCIPAL DISPLAY PANEL - 500 mg - 10 ct. Blister Card - zithromax 11
- PRINCIPAL DISPLAY PANEL - 500 mg Tablet Blister Pack Box - zithromax 12
- Principal Display Panel - 300 mg Bottle Label - zithromax 13
- PRINCIPAL DISPLAY PANEL - 300 mg Bottle Carton - zithromax 14
- PRINCIPAL DISPLAY PANEL - 600 mg Bottle Label - zithromax 15
- PRINCIPAL DISPLAY PANEL - 600 mg Bottle Carton - zithromax 16
- PRINCIPAL DISPLAY PANEL - 900 mg Bottle Label - zithromax 17
- PRINCIPAL DISPLAY PANEL - 900 mg Bottle Carton - zithromax 18
- PRINCIPAL DISPLAY PANEL - 1200 mg Bottle Label - zithromax 19
- PRINCIPAL DISPLAY PANEL - 1200 mg Bottle Carton - zithromax 20
Product Label Images
The following 21 images provide visual information about the product associated with Zithromax NDC 0069-3130 by Pfizer Laboratories Div Pfizer Inc, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Logo - zithromax 02

This is a brief description indicating that the product is distributed by Pfizer Labs, which is a division of Pfizer Inc located in New York City.*
zithromax 02a

"Distributed by Pfizer Labs, Division of Pfizer Inc, NY, NY 10017" - This text provides information regarding the distribution of pharmaceutical products by Pfizer Labs, which is a division of Pfizer Inc. The stated address is situated in New York with the zip code being 10017.*
PRINCIPAL DISPLAY PANEL - 250 mg - 30 Tablet Bottle Label - zithromax 03

This text is a description of the medication Zithromax (azithromycin) tablets. It includes dosage and use information, the NDC and GTIN codes, and advises to store the medicine at a temperature between 15° and 30°C in child-resistant containers. The medication is dispensed in tight (USP) containers and each tablet contains azithromycin difydrate equivalent to 250 mg of azithromycin. The medicine is distributed by Pfizer Labs and the package contains 30 tablets and is for prescription use only.*
Principal Display Panel - 250 mg - 6 ct. Blister Pack - zithromax 04

This is an advertisement for Zithromax, an antibiotic for treating bacterial infections. The advertisement suggests taking two tablets on the first day followed by one tablet daily for the next four days. It claims that the drug keeps on working, and a full course of therapy is recommended. The text also mentions that Zithromax costs less compared to other antibiotics. The copy also contains some random errors and symbols, but the overall meaning and message of the text are understandable.*
PRINCIPAL DISPLAY PANEL - 250 mg Tablet Blister Pack Box - zithromax 07

Zithromax (azithromycin) tablets in a bottle with 50 tablets. The package is a unit dose and is intended for use in a medical setting only. Each tablet is equivalent to 50mg of azithromycin. The manufacturer's address is Do, W, 087.*
PRINCIPAL DISPLAY PANEL - 500 mg - 30 Tablet Bottle Label - zithromax 08

This text provides information on how to store and dispense Zithromax tablets which contain azithromycin dihydrate. The tablets must be stored between 15 and 0 degrees Celsius (59 to 86 degrees Fahrenheit) and dispensed tightly according to USP standards. The dosage and usage instructions can be found in the accompanying prescribing information. Each tablet contains 500g of azithromycin dihydrate. The manufacturer is Pfizer Labs, a division of Pz Inc, located in NY, USA. The product is distributed in a 30-tablet package that is available only by prescription. The LOT number and expiration date are not available in the provided text.*
Principal Display Panel - 500 mg - 3 ct. Tablet Blister Pack - zithromax 09

Zithromax is an oral antibiotic that is used to treat bacterial infections. It is taken once a day for three days. Zithromax tablets can be taken with or without food. The most common side effects of Zithromax are diarrhea, nausea, and stomach pain. In rare cases of an allergic reaction, you should stop taking this drug immediately and call your healthcare provider. If you have any questions about this drug or its possible side effects, please talk to your healthcare provider.*
PRINCIPAL DISPLAY PANEL - 500 mg - 10 ct. Blister Card - zithromax 11

Zithromax® (azithromycin) Tablet is available in 500mg and is distributed by Pfizer labs. The text contains some errors and repetitions.*
PRINCIPAL DISPLAY PANEL - 500 mg Tablet Blister Pack Box - zithromax 12
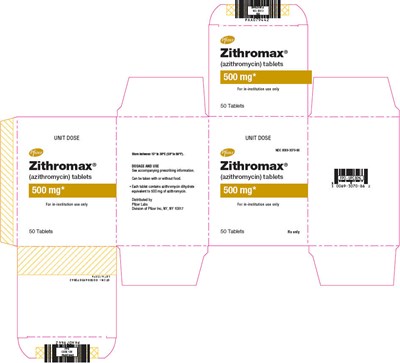
Zithromax® (azithromycin) tablets are available in a unit dose package containing 50 tablets of 500mg each. These tablets are for informational use only and are not intended for treatment purposes.*
Principal Display Panel - 300 mg Bottle Label - zithromax 13

This appears to be instructions for a medication called Zithromax containing 300 mg of azithromycin. The medication comes in a dry powder form that should be stored below 30°C. To take the medication, 9 mL of water needs to be added to the bottle and then shaken well. The dosage and use instructions should be followed as provided in the accompanying prescription information. The medication should be used within 10 days of mixing and provides extra space for shaking. The medication should be discarded after use and should be protected from freezing. The medication is cherry-flavored and for oral use only.*
PRINCIPAL DISPLAY PANEL - 300 mg Bottle Carton - zithromax 14

This is a description of Zithromax, an oral suspension medication containing azithromycin. It is a cherry-flavored powder that, when mixed with water, provides 100 mg of medication per 5 mL. The medication comes in an oversized bottle, and the label on the bottle advises shaking well before use. Proper storage involves keeping it at temperatures below 30°C (86°F) and avoiding freezing. The dosage and usage instructions are available with the accompanying prescription. The text also includes a phone number and address for queries about the medication.*
PRINCIPAL DISPLAY PANEL - 600 mg Bottle Label - zithromax 15

This text is a set of instructions and precautions for the oral use of Zithromax, an oral suspension azithromycin medication. The bottle contains dry powder that should be stored below 30°C and protected from freezing. The mixing directions indicate adding 9mL of water to the bottle to create a cherry-flavored oral suspension containing 200mg per 5mL. The bottle is oversized to provide extra space for shaking. The medication is for prescription use only, and the instructions advise shaking well before using and discarding all dosages once completed.*
PRINCIPAL DISPLAY PANEL - 600 mg Bottle Carton - zithromax 16
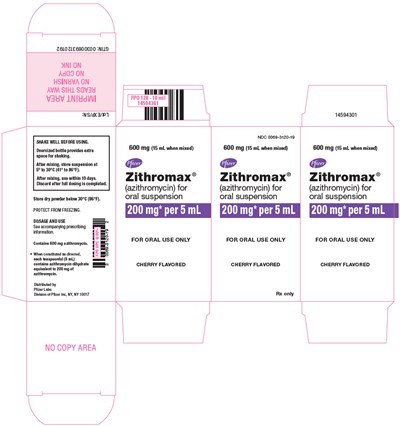
This appears to be a description of a medication called Zithromax, which is available in an oral suspension form containing 600mg of azithromycin. The suspension is cherry flavored and should be shaken well before use. The dosage and usage information should be followed according to the accompanying prescription information. When constituted, each teaspoonful contains 200mg of azithromycin, with enough suspension to contain 600mg total. The medication should be stored below 30°C and protected from freezing. The manufacturer's address is also listed.*
PRINCIPAL DISPLAY PANEL - 900 mg Bottle Label - zithromax 17

Zithromax, also known as azithromycin, is a dry powder medication that must be stored at temperatures below 30°C (86°F) and protected from freezing. The mixing directions involve combining 1200mg of the powder with 12 mL of water in the bottle. After shaking the mixture, the suspension can be used within 10 days and the UL closing should be discarded. The medication is cherry-flavored with a dosage of 200 mg per 5 mL of mixed solution. The product is produced by Pfizer and has a unique identification number of NOG 0069-9130-19.*
PRINCIPAL DISPLAY PANEL - 900 mg Bottle Carton - zithromax 18

This is a prescription for Zithromax oral suspension which contains 900 mg atvomycin for oral use only. It is cherry flavored and should be shaken well before use. The recommended dosage and usage information should be obtained from the accompanying prescribing information. It must be stored in dry powder below 0°C, and protected from freezing. Each teaspoonful contains 20 mg of Seitromein.*
PRINCIPAL DISPLAY PANEL - 1200 mg Bottle Label - zithromax 19

This text is a set of instructions for using Zithromax oral suspension, which contains azithromycin. It must be stored below 30°C and protected from freezing. The dosage and mixing directions are included, and users must shake the mixture well before use. The bottle should be used within ten days of mixing, and the enclosed dosing information should be referred to. The suspension is cherry flavored and contains 1200mg of azithromycin. The distributor is Pfizer Labs.*
PRINCIPAL DISPLAY PANEL - 1200 mg Bottle Carton - zithromax 20

This is a medication for oral use only, with 1200mg of azithromycin in cherry-flavored suspension form. The drug needs to be stored at room temperature and protected from freezing. The prescription information should be read before use. Shake the bottle well before using it. There are different variations of dosages depending on the prescription. The medication is manufactured by a pharmaceutical company named Pfizer.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.



