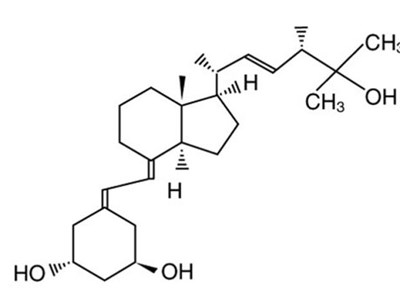
Product Images Zemplar
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 13 images provide visual information about the product associated with Zemplar NDC 0074-4637 by Abbvie Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
zemplar injection spl 02

Zemplar® (paicalcitol) Injection is a medication available at a concentration of 2 meg/ml. The rest of the text contains limited and unclear information and may not be useful.*
zemplar injection spl 03

This is not a readable text. It seems to be a partially recognized label of an injection called "Zemplar".*
zemplar injection spl 05

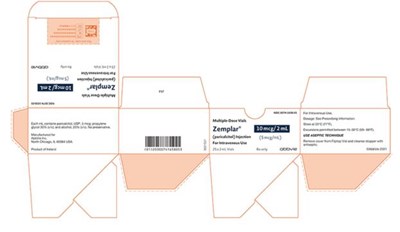
The description is: Zemplar® (paricalcitol) Injection is a medication that comes in 28 mL vials for intravenous use. The package has a NOC number of NOC00741658.01 and is manufactured by Abbvie. There is additional text, but it is not readable through .*
zemplar injection spl 07

Zemplar is a paricalcitol injection that comes in 25x2mL vials for intravenous use with an NDC number of 0074-165805 and labeled as RX only. The remaining text is not legible.*
zemplar injection spl 09

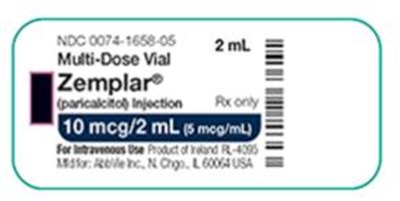
This is a description of a medication called Zemplar. It comes in a multiple-dose vial size of 2 mL with the code NOC 0074-1656-05. The injection contains 10 mcg/2mL and 5 meg of paricalcitol for intravenous use. The lot number is 8021424 but the expiration date is not provided.*
zemplar injection spl 0b

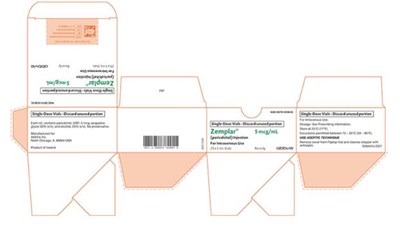
This is a description of a medication in a single-dose vial. The medication is called Zemplar and it is an injection used for intravenous use. The vial contains 1 milliliter of the medication with a concentration of 5 milligrams per milliliter. The manufacturer of the medication is AbbVie Inc. The text also includes a code number "NOCOO74-1658.01" and instructions to discard any unused portion.*
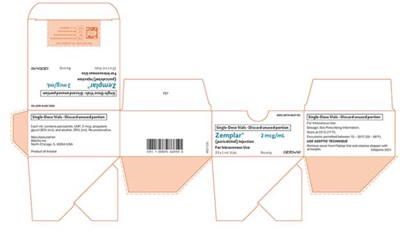
zemplar injection spl 0d

The text is a description of Zemplar® Injection, a medication containing paricalcitol in a single-dose vial of 1mL and a concentration of 2 meg/mL for intravenous use only. The label includes the National Drug Code (NDC) and indicates that this medication is available only by prescription. The manufacturer is identified as Abbie Inc, located in Chicago, IL, USA, with the product originating from Ireland. The label also includes a unique identifier number (8021428) and instructions to discard any unused portion of the medication.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.