Product Images Depakote Sprinkles
View Photos of Packaging, Labels & Appearance
- Divalproex sodium is a stable co-ordination compound comprised of sodium valproate and valproic acid in a 1:1 molar relationship and formed during the partial neutralization of valproic acid with 0.5 equivalent of sodium hydroxide. Chemically it is designated as sodium hydrogen bis(2-propylpentanoate). Divalproex sodium has the following structure: - depakote sprinkle capsules spl 01
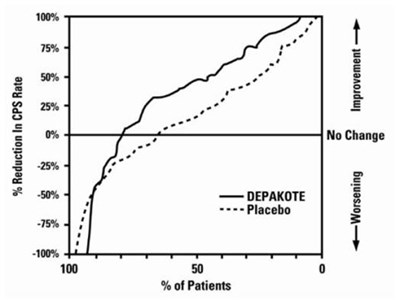
- Figure 1 - depakote sprinkle capsules spl 02
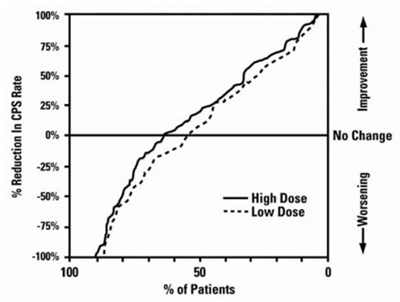
- Figure 2 - depakote sprinkle capsules spl 03
- 1 - depakote sprinkle capsules spl 04
- 2 - depakote sprinkle capsules spl 05
- 3 - depakote sprinkle capsules spl 06
- 4 - depakote sprinkle capsules spl 07
- NDC 0074–6114–13 100 Sprinkle Capsules DEPAKOTE® SPRINKLE CAPSULES Divalproex Sodium Delayed Release Capsules 125 mg Valproic Acid Activity Dispense the accompanying Medication Guide to each patient. Rx only abbvie - depakote sprinkle capsules spl 08
- Description: delisted image - depakote sprinkle capsules spl 09
Product Label Images
The following 9 images provide visual information about the product associated with Depakote Sprinkles NDC 0074-6114 by Abbvie Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Divalproex sodium is a stable co-ordination compound comprised of sodium valproate and valproic acid in a 1:1 molar relationship and formed during the partial neutralization of valproic acid with 0.5 equivalent of sodium hydroxide. Chemically it is designated as sodium hydrogen bis(2-propylpentanoate). Divalproex sodium has the following structure: - depakote sprinkle capsules spl 01