Product Images Entresto
View Photos of Packaging, Labels & Appearance
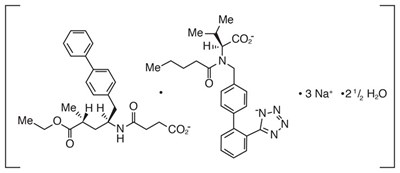
- Valsartan structural formula - ENTRESTO 01
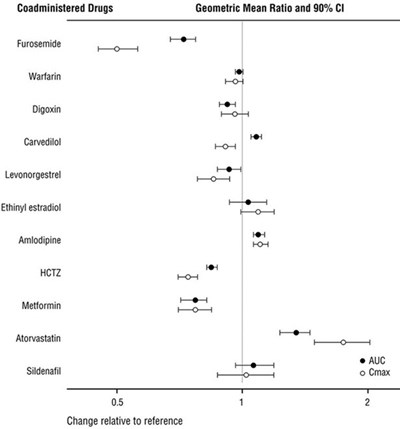
- Figure 1: Effect of ENTRESTO on Pharmacokinetics of Coadministered Drugs - ENTRESTO 02
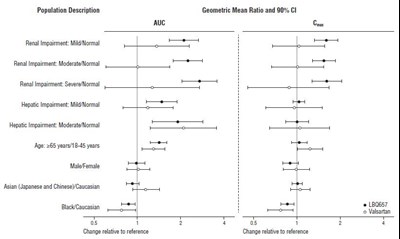
- Figure 2: Pharmacokinetics of ENTRESTO in Specific Populations - ENTRESTO 03
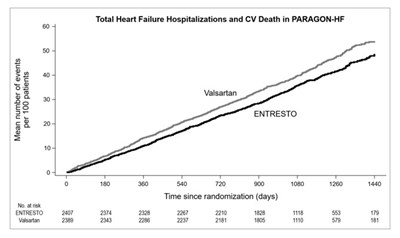
- Figure 3: Kaplan-Meier Curves for the Primary Composite Endpoint (A), Cardiovascular Death (B), and Heart Failure Hospitalization (C) - ENTRESTO 04
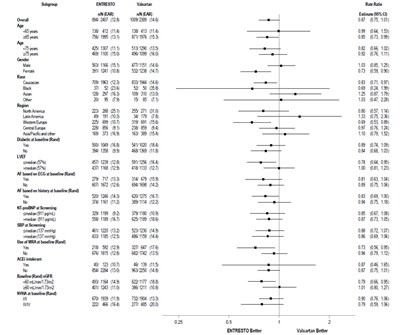
- Figure 4: Primary Composite Endpoint (CV Death or HF Hospitalization) - Subgroup Analysis - ENTRESTO 05
- ENTRESTO 06
- ENTRESTO 07
- ENTRESTO 08
- ENTRESTO 09
- ENTRESTO 10
- ENTRESTO 11
- ENTRESTO 12
- ENTRESTO 13
Product Label Images
The following 13 images provide visual information about the product associated with Entresto NDC 0078-0659 by Novartis Pharmaceuticals Corporation, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Figure 3: Kaplan-Meier Curves for the Primary Composite Endpoint (A), Cardiovascular Death (B), and Heart Failure Hospitalization (C) - ENTRESTO 04
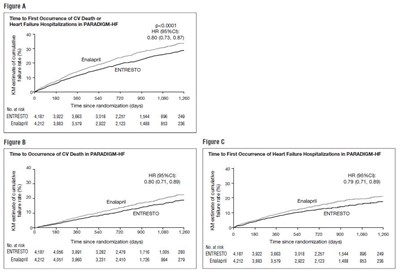
The text appears to be a comparison of time to the first occurrence of heart failure hospitalizations or CV death in patients treated with Entresto in the PARADIGNHE study. The data is presented in two figures, A and B, graphing the occurrence over time since randomization.*
Figure 4: Primary Composite Endpoint (CV Death or HF Hospitalization) - Subgroup Analysis - ENTRESTO 05
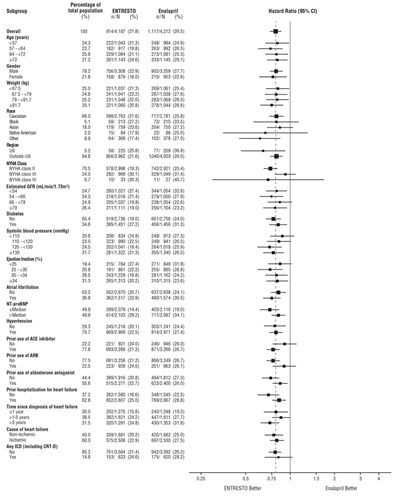
This appears to be a table presenting clinical data from a study with two groups receiving different medications (Entresto and Enalapril). The data is organized based on different patient subgroups according to age, gender, weight, race, NYHA class, diabetes, atrial fibrillation and other factors. The table presents the total number and percentage of patients in each subgroup, as well as the percentage of patients in each group that displayed better results or a better outcome. A hazard ratio (95%CI) is also presented, but it is not clear from the information given what it refers to.*
ENTRESTO 06
This is a description of the drug called Entresto, manufactured by Novartis. It is a prescription tablet made of sacubitril/valsartan and is used to treat individuals with heart failure. The recommended storage temperature for this medication is between 20°C to 25°C. This text also includes the manufacturer's information and the distributor's details.*
ENTRESTO 07

Entresto® is a medication available in tablet form with a strength of 60 tablets per prescription. The manufacturer's recommended dosage should be followed, and the medication should be stored at controlled room temperature between 20°C to 25°C (88°F to 71°F) with variations allowed between 15°C to 30°C (59°F to 86F) as controlled by USP. The medication should be kept away from moisture and out of reach of children. Contact the manufacturer at 1-888-838-ENTRESTO (1-888-9687378) or visit their website for more information.*
ENTRESTO 08
Entresto is a prescription drug available in tablet form. The tablets contain sacubitril and valsartan and are available in the strength of 97mg/103mg. The recommended storage temperature is between 20°C to 25°C (68°F t0 71°F) with excursions permitted at 15°C to 30°C (69°F to 85°F). It's important to protect the tablets from moisture and keep them out of reach of children. For more information on usage and dosage, consult with a prescribing physician. For more support, call 888 ENTRESTO (1-8883607378). This drug is manufactured by Novartis Pharmaceuticals.*
ENTRESTO 11
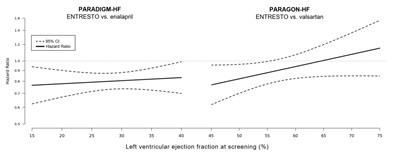
The text above shows a table with two columns, representing Hazard Ratio and Left Ventricular Ejection Fraction at screening in percentage (%). The table shows the Hazard Ratio results comparing ENTRESTO versus enalapril and ENTRESTO versus valsartan in two clinical trials named PARADIGM-HF and PARAGON-HF. The table displays values ranging from 16 to 0.5.*
ENTRESTO 12

This is a sample pack of Entresto medication, which is a combination of sacubitril and valsartan used to treat heart failure. The pack includes 28 tablets of 24mg/26mg strength, and is only available with a prescription from a physician. The pack is not for sale, and its purpose is to allow physicians to familiarize themselves with the medication before prescribing it to their patients. The medication is produced by Novartis.*
ENTRESTO 13

This is a physician sample of Entresto (sacubitril valsartan) with a dosage of 49mg/51mg. The sample contains 28 tablets and is only available for prescription use. The medication is manufactured by Novartis and should not be sold.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.