Product Images Mekinist
View Photos of Packaging, Labels & Appearance
- Trametinib Structure-01 - mekinist 01
- Figure 1. Kaplan-Meier Curves of Investigator-Assessed Progression-Free Survival (ITT Population) in the METRIC Study - mekinist 02
- Figure 2. Kaplan Meier Curves of Overall Survival in the COMBI-d Study - mekinist 03
- mekinist 04
- mekinist 05
- mekinist 06
- mekinist 07
- mekinist 08
Product Label Images
The following 8 images provide visual information about the product associated with Mekinist NDC 0078-1112 by Novartis Pharmaceuticals Corporation, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Figure 1. Kaplan-Meier Curves of Investigator-Assessed Progression-Free Survival (ITT Population) in the METRIC Study - mekinist 02

Figure 2. Kaplan Meier Curves of Overall Survival in the COMBI-d Study - mekinist 03
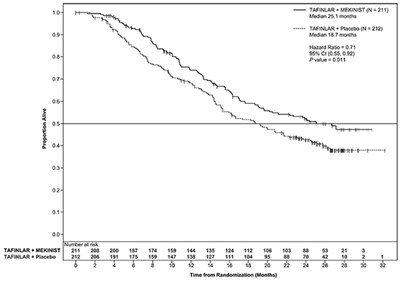
This is a comparison of TAFINLAR + MEKINIST versus TAFINLAR + Placebo in a study involving 211 and 212 people, respectively. The median progression-free survival for TAFINLAR + MEKINIST was 25.1 months versus 18.7 months for TAFINLAR + Placebo. The Hazard Ratio was 0.71 with a 95% CI of 0.55-0.92 and the P-value was 0.001. The data is presented in a graph.*
mekinist 04

This is a medication called Mekinist that comes in the form of tablets. The package contains 30 tablets, and each tablet has 0.5 mg of trametinib. The recommended dosage is not given in the text and should be obtained from the prescribing information. The medication is manufactured by Glaxosmithiine and distributed by Novartis. It should be stored refrigerated at 2°C to 8°C and protected from moisture and light. The safety seal under the cap should not be broken or missing, and the medication should not be placed in pillboxes. The NDC number for this medication is 0078-0666-15.*
mekinist 05

This is a description of Trametinib tablets with a dosage of 2254mg and 2mg of Rametinb. There are instructions to see accompanying prescribing information and to store the tablets refrigerated at 2°C08°C (36°F to 46°F). The product is made by Glaxosmithkline Manufacturing at their location in Singapore, and distributed by Novartis in East Hanover, NJ. There are warnings not to use the tablets if the printed safety seal is broken or missing, not to remove the desiccant, and not to place medication in pill boxes. However, some parts of the text are not readable, hence the information in the text is limited.*
mekinist 06
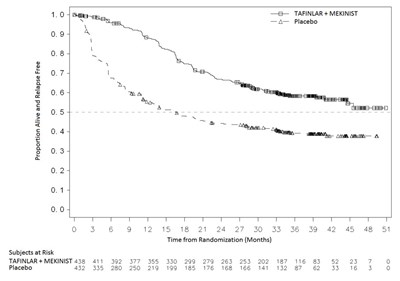
This appears to be a chart or graph with two groups (TAFINLAR + MEKINIST and Placebo) plotted against time in months. The chart displays the number of subjects at risk in each group for specific time intervals. However, without additional context, it is unclear what the data represents.*
mekinist 07

This is a medication description for Mekinist (trametinib) tablets. The tablets contain 0.5 mg of trametinib and dimethyl sulfoxide. The dosage instructions are included in the prescribing information. The medication is manufactured by Novartis Pharma Stein AG in Switzerland and distributed in the United States by Novartis in East Hanover, NJ. The product must be stored refrigerated and protected from light and moisture. The tablets should not be removed from the original bottle or placed in pillboxes.*
mekinist 08

This is a description of a medication called Mekinist, which comes in the form of tablets containing 2254 mg of trametinib and dimethyl sulfide equivalent to 102 mg of trametinib. The dosage and prescribing information can be found in the accompanying documentation. The manufacturer is Novartis Pharma Stin AG, and the product is distributed by Novartis in East Hanover, NJ. The tablets should be stored in a refrigerated environment between 2°C and 8°C and protected from moisture and light. It is important not to remove the desiccant or place the pills in pill boxes.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.
