Product Images Fluticasone Propionate And Salmeterol
View Photos of Packaging, Labels & Appearance
- FP chem structure - fluticasone propionate salmeterol inhalation powde 1
- Figure 8 - fluticasone propionate salmeterol inhalation powde 10
- Teva Logo - fluticasone propionate salmeterol inhalation powde 11
- Teva Logo - fluticasone propionate salmeterol inhalation powde 12
- Figurr A - fluticasone propionate salmeterol inhalation powde 13
- Figure B - fluticasone propionate salmeterol inhalation powde 14
- Figure C - fluticasone propionate salmeterol inhalation powde 15
- Figure D - fluticasone propionate salmeterol inhalation powde 16
- Breathe out image - fluticasone propionate salmeterol inhalation powde 17
- Figure F - fluticasone propionate salmeterol inhalation powde 18
- Figure G - fluticasone propionate salmeterol inhalation powde 19
- Salmeterol chem structure - fluticasone propionate salmeterol inhalation powde 2
- Figure H - fluticasone propionate salmeterol inhalation powde 20
- Teva Logo - fluticasone propionate salmeterol inhalation powde 21
- NDC 0093-3607-82 - Carton - 55 mcg / 14 mcg - fluticasone propionate salmeterol inhalation powde 22
- 113 mcg/ 14 mcg Carton - NDC 0093-3608-82 - fluticasone propionate salmeterol inhalation powde 23
- 232 mcg / 14 mcg Carton - NDC 0093-3609-82 - fluticasone propionate salmeterol inhalation powde 24
- Figure 1 - fluticasone propionate salmeterol inhalation powde 3
- Figure 2 - fluticasone propionate salmeterol inhalation powde 4
- Figure 3 - fluticasone propionate salmeterol inhalation powde 5
- Figure 4 - fluticasone propionate salmeterol inhalation powde 6
- Figure 5 - fluticasone propionate salmeterol inhalation powde 7
- Figure 6 - fluticasone propionate salmeterol inhalation powde 8
- Figure 7 - fluticasone propionate salmeterol inhalation powde 9
Product Label Images
The following 24 images provide visual information about the product associated with Fluticasone Propionate And Salmeterol NDC 0093-3609 by Teva Pharmaceuticals Usa, Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
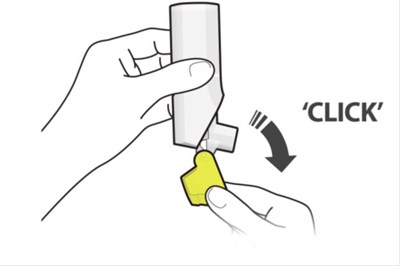
Figurr A - fluticasone propionate salmeterol inhalation powde 13

The text describes the features of a medical device, which includes a dose counter, a vent, a mouthpiece, and a yellow cap. It is most likely an inhaler or similar product used for administering medication.*
Figure B - fluticasone propionate salmeterol inhalation powde 14

This is a description of an inhaler that has 60 doses when full and is indicated as empty when it has no doses left.*
NDC 0093-3607-82 - Carton - 55 mcg / 14 mcg - fluticasone propionate salmeterol inhalation powde 22

This is a description of a medication called "Fluticasone Propionate and Salmeterol Inhalation Powder." The medication is meant for oral inhalation and includes a dose counter. The packaging indicates a net content of 0.45 g and 60 metered inhalations. The enclosed patient leaflet contains information on product use and potential side effects.*
113 mcg/ 14 mcg Carton - NDC 0093-3608-82 - fluticasone propionate salmeterol inhalation powde 23

This text describes a medication called "Fluticasone Propionate and Salmeterol Inhalation Powder" that is used for oral inhalation. It comes with a dose counter and provides 60 metered inhalations per 0.45g net contents. The user is directed to refer to the enclosed patent leaflet and instructions for use for information on product usage and handling.*
232 mcg / 14 mcg Carton - NDC 0093-3609-82 - fluticasone propionate salmeterol inhalation powde 24

This appears to be the packaging and labeling information for a medication called "Fluticasone Propionate and Salmeterol Inhalation Powder." It is for oral inhalation only, has a dose counter, and comes with a patient leaflet. The net contents are 0.45 g and there are 60 metered inhalations. The medication contains 232 mcg of fluticasone propionate and 14 mcg of salmeterol.*
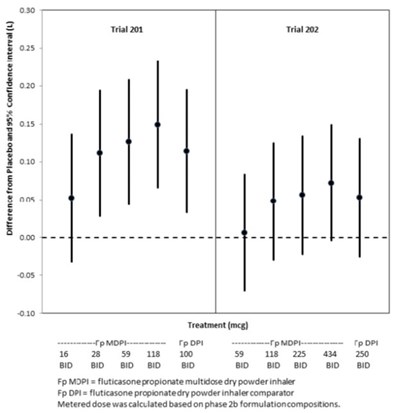
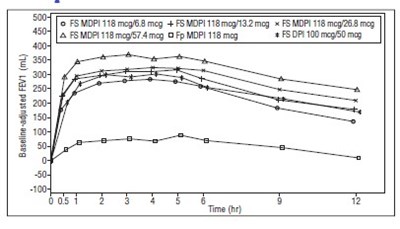
Figure 4 - fluticasone propionate salmeterol inhalation powde 6
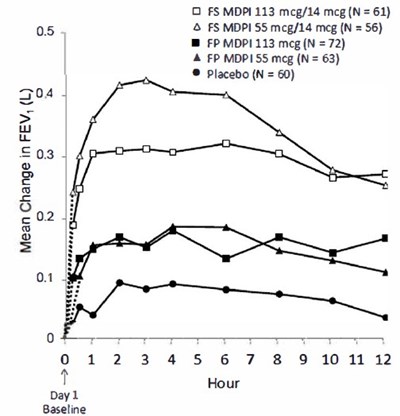
This appears to be a table or chart containing information about different medications and their effectiveness in improving FEV (forced expiratory volume) over time. The medications listed are O FS MDPI, 05 & FS MDPI, FP MDPI, A FP MDP15S, and a placebo. The number of participants (N) in each group is also given. However, without additional context or clarification, it is difficult to provide a more detailed or specific description of the text.*
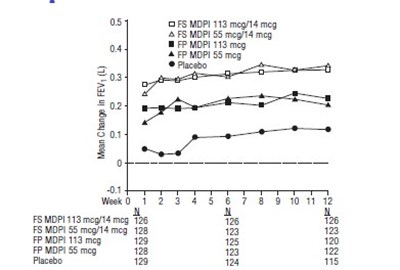
Figure 7 - fluticasone propionate salmeterol inhalation powde 9
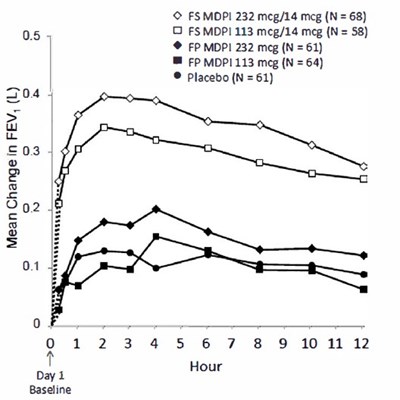
This text represents a table showing the mean change in FEV (forced expiratory value) measured in liters. The table compares the change in FEV of different doses of FSMDPI and MDPI with a placebo over time. However, it is unclear what the units on the horizontal axis represent.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.